Compounds Are Represented By Chemical Formulas
Kalali
Apr 05, 2025 · 7 min read
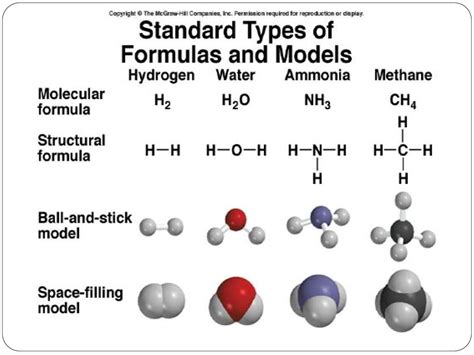
Table of Contents
Compounds are Represented by Chemical Formulas: A Deep Dive into Chemical Notation
Chemical formulas are the cornerstone of chemistry, providing a concise and standardized way to represent the composition of chemical compounds. Understanding chemical formulas is crucial for anyone studying chemistry, from beginners to seasoned professionals. This comprehensive guide delves into the intricacies of chemical formulas, exploring their different types, how they are constructed, and their significance in various chemical contexts. We'll unravel the secrets behind these symbolic representations and explore how they unlock a deeper understanding of the chemical world.
Understanding the Basics: What is a Chemical Formula?
A chemical formula is a symbolic representation of the elements and their proportions within a chemical compound. It provides crucial information about the types of atoms present and their relative numbers. This information is vital for various applications, including predicting reactions, balancing equations, and understanding the properties of the compound. For example, the chemical formula for water, H₂O, tells us that each molecule of water contains two hydrogen atoms and one oxygen atom.
Key Components of a Chemical Formula:
- Element Symbols: These are one or two-letter abbreviations for the elements (e.g., H for hydrogen, O for oxygen, C for carbon). These symbols are universally recognized by scientists worldwide.
- Subscripts: These are small numbers written slightly below and to the right of an element's symbol. They indicate the number of atoms of that element present in one molecule or formula unit of the compound. If no subscript is written, it is understood to be 1.
Types of Chemical Formulas: A Variety of Representations
Several types of chemical formulas exist, each offering a slightly different perspective on the composition and structure of a compound. Understanding these variations is essential for interpreting chemical information effectively.
1. Empirical Formula: The Simplest Representation
The empirical formula shows the simplest whole-number ratio of atoms of each element in a compound. It doesn't necessarily represent the actual number of atoms in a molecule, but rather the ratio between them. For example, the empirical formula for glucose is CH₂O, even though the actual molecular formula is C₆H₁₂O₆. This means that for every carbon atom, there are two hydrogen atoms and one oxygen atom. Empirical formulas are often determined experimentally through techniques like elemental analysis.
2. Molecular Formula: Reflecting the Actual Composition
The molecular formula shows the actual number of atoms of each element present in a single molecule of a compound. It provides a complete picture of the molecular composition. As mentioned earlier, the molecular formula for glucose is C₆H₁₂O₆, which accurately represents the six carbon atoms, twelve hydrogen atoms, and six oxygen atoms in each glucose molecule. The molecular formula is a multiple of the empirical formula.
3. Structural Formula: Visualizing the Arrangement of Atoms
The structural formula goes beyond simply showing the number and type of atoms. It visually represents the arrangement of atoms within a molecule, showing the bonds between them. This allows for a better understanding of the molecule's shape and properties. For example, the structural formula for ethanol (C₂H₅OH) shows the arrangement of the carbon, hydrogen, and oxygen atoms and the bonds connecting them, illustrating the presence of a hydroxyl group (-OH). This visual representation is crucial for understanding the reactivity and properties of the molecule.
4. Condensed Structural Formula: A More Compact Representation
Condensed structural formulas are a more compact way of representing the structure of a molecule. They still show the connections between atoms but do so in a more streamlined way. For example, the condensed structural formula for ethanol is CH₃CH₂OH, omitting the explicit representation of each bond but still clearly indicating the connectivity of the atoms. This format is often used for larger molecules where the full structural formula would be cumbersome.
5. Skeletal Formula (Line-Angle Formula): A Simplified Representation
Skeletal formulas, also known as line-angle formulas, are the most simplified representation of organic molecules. Carbon atoms are implied at the intersections and ends of lines, and hydrogen atoms attached to carbon atoms are not explicitly shown. Other atoms (like oxygen, nitrogen, etc.) are explicitly shown. This simplification makes it easier to represent complex organic molecules while maintaining the crucial information about their structure.
Ionic Compounds: Representing Ions and Charges
Ionic compounds are formed through the electrostatic attraction between positively charged ions (cations) and negatively charged ions (anions). Their chemical formulas represent the simplest whole-number ratio of these ions, reflecting the electrical neutrality of the compound. For example, sodium chloride (NaCl) has a 1:1 ratio of sodium cations (Na⁺) and chloride anions (Cl⁻), indicating that the positive and negative charges balance out.
Polyatomic Ions: Groups of Atoms with a Charge
Polyatomic ions are groups of atoms that carry a net electric charge. These ions are treated as single units in chemical formulas. For example, the sulfate ion (SO₄²⁻) is a polyatomic ion with a charge of -2. In the chemical formula for magnesium sulfate (MgSO₄), the magnesium cation (Mg²⁺) balances the charge of the sulfate ion.
Hydrates: Incorporating Water Molecules
Hydrates are compounds that contain water molecules incorporated into their crystal structure. The number of water molecules per formula unit is indicated by a dot followed by the number and the formula for water (H₂O). For example, copper(II) sulfate pentahydrate is represented as CuSO₄·5H₂O, indicating five water molecules are associated with each formula unit of copper(II) sulfate.
The Importance of Chemical Formulas: Applications and Significance
Chemical formulas are not merely symbolic representations; they are essential tools in various chemical disciplines. Their significance extends across diverse applications:
- Stoichiometry: Chemical formulas are fundamental to stoichiometry, the quantitative study of chemical reactions. They allow us to calculate the amounts of reactants and products involved in a reaction based on the balanced chemical equation.
- Naming Compounds: The system for naming chemical compounds (nomenclature) is directly based on chemical formulas. By analyzing the formula, one can determine the correct name for a compound and vice versa.
- Predicting Properties: The chemical formula provides insights into the physical and chemical properties of a compound. For example, the presence of certain functional groups indicated by the formula can help predict reactivity.
- Understanding Chemical Reactions: Chemical formulas are crucial for understanding and writing balanced chemical equations, which describe the rearrangement of atoms during a chemical reaction.
- Laboratory Work: In laboratory settings, chemical formulas are essential for preparing solutions, identifying substances, and carrying out various chemical experiments.
Advanced Concepts: Beyond Basic Formulas
While the basic types of chemical formulas cover most situations, advanced concepts require a deeper understanding. These include:
- Isomers: Molecules with the same molecular formula but different structural arrangements are called isomers. Isomers can have vastly different properties even though they have the same chemical formula.
- Coordination Compounds: Coordination compounds involve metal ions surrounded by ligands (molecules or ions). Their formulas often utilize square brackets to enclose the coordination sphere.
- Organic Chemistry: Organic chemistry relies heavily on structural formulas to understand the immense variety and complexity of carbon-containing molecules. Different notations, like condensed and skeletal formulas, are commonly used to efficiently represent organic compounds.
Conclusion: Unlocking the Secrets of Chemical Composition
Chemical formulas are more than just symbols; they are powerful tools that provide a concise yet comprehensive representation of the composition and structure of chemical compounds. By understanding the various types of formulas and their applications, one gains a deeper appreciation for the fundamental principles of chemistry. From simple empirical formulas to complex structural representations, these symbolic notations serve as the foundation for understanding the chemical world around us and unlock the secrets of matter's composition. Mastering chemical formulas is essential for anyone seeking to delve deeper into the fascinating realm of chemistry.
Latest Posts
Latest Posts
-
Can A Sedimentary Rock Become An Igneous Rock
Apr 05, 2025
-
How Many Feet Is 104 In
Apr 05, 2025
-
How Tall Is 54 In In Feet
Apr 05, 2025
-
How Long Is 34 Cm In Inches
Apr 05, 2025
-
70 Cm Equals How Many Inches
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Compounds Are Represented By Chemical Formulas . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
