Do Covalent Bonds Give Water A Low Heat Capacity
Kalali
Apr 03, 2025 · 5 min read
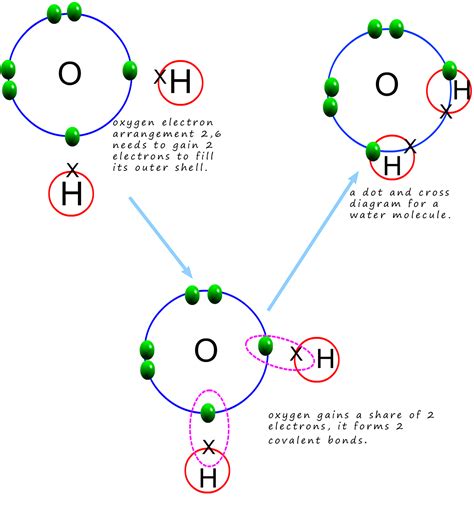
Table of Contents
Do Covalent Bonds Give Water a Low Heat Capacity? A Deep Dive into Water's Unique Properties
Water. The elixir of life. A seemingly simple molecule, yet its properties are anything but. One of water's most remarkable characteristics is its exceptionally high specific heat capacity – the amount of heat required to raise the temperature of one gram of a substance by one degree Celsius. This high heat capacity is crucial for regulating Earth's climate and supporting life as we know it. But what causes this seemingly counterintuitive property? The common misconception is that the covalent bonds within the water molecule are responsible for its low heat capacity. This is incorrect. In fact, it's the opposite: water's high heat capacity is a direct consequence of its unique molecular structure and the nature of its hydrogen bonds, not its covalent bonds. Let's delve into the science to understand why.
Understanding Covalent Bonds in Water
Water (H₂O) is a molecule formed by two hydrogen atoms covalently bonded to a single oxygen atom. A covalent bond is a chemical bond formed when two atoms share one or more pairs of electrons. In water, the oxygen atom shares electrons with each hydrogen atom, creating a stable molecule. These covalent bonds are relatively strong, requiring a significant amount of energy to break. However, the strength of these covalent bonds is not the primary factor determining water's high heat capacity. While the covalent bonds contribute to the overall stability of the water molecule, they do not directly explain its high heat capacity.
The Role of Hydrogen Bonds in Water's High Heat Capacity
The key to understanding water's high heat capacity lies in the hydrogen bonds formed between water molecules. Hydrogen bonds are a special type of intermolecular force (a force between molecules, not within a molecule) that occurs when a hydrogen atom bonded to a highly electronegative atom (like oxygen or nitrogen) is attracted to another electronegative atom in a nearby molecule.
In water, the oxygen atom is more electronegative than the hydrogen atoms, resulting in a polar molecule. The oxygen atom carries a partial negative charge (δ-), and the hydrogen atoms carry partial positive charges (δ+). This polarity allows water molecules to form hydrogen bonds with each other. Each water molecule can form up to four hydrogen bonds with neighboring water molecules, creating a complex three-dimensional network.
How Hydrogen Bonds Influence Heat Capacity
This extensive hydrogen bonding network is responsible for water's high heat capacity. When heat is added to water, the energy is initially used to break these hydrogen bonds, not to significantly increase the kinetic energy (and thus the temperature) of the water molecules. A substantial amount of energy is required to disrupt these bonds because they are relatively strong compared to other intermolecular forces. Consequently, a large amount of heat energy is absorbed with a relatively small increase in temperature. Conversely, when heat is removed from water, hydrogen bonds reform, releasing energy and slowing down the rate of temperature decrease.
This is in stark contrast to substances with weaker intermolecular forces. In these substances, adding heat directly increases the kinetic energy of the molecules, leading to a more rapid increase in temperature. Therefore, these substances exhibit lower heat capacities.
Comparing Water's Heat Capacity to Other Substances
To fully appreciate water's unique heat capacity, let's compare it to other common substances:
- Water (H₂O): Specific heat capacity ≈ 4.18 J/g°C
- Ethanol (C₂H₅OH): Specific heat capacity ≈ 2.44 J/g°C
- Ammonia (NH₃): Specific heat capacity ≈ 4.71 J/g°C
- Methane (CH₄): Specific heat capacity ≈ 2.20 J/g°C
As we can see, water's specific heat capacity is significantly higher than many other common liquids. This is directly attributable to the strong hydrogen bonding network present in liquid water. While ammonia also displays hydrogen bonding, its lower heat capacity compared to water points to the importance of the specific arrangement and number of hydrogen bonds in influencing heat capacity.
Consequences of Water's High Heat Capacity
Water's exceptionally high heat capacity has profound implications for both the Earth's climate and the existence of life:
Climate Regulation:
-
Moderating Temperature Fluctuations: Large bodies of water, such as oceans and lakes, absorb vast amounts of solar energy without experiencing drastic temperature changes. This helps to moderate global temperatures and prevent extreme temperature fluctuations, creating a more stable environment. Coastal regions, for instance, experience smaller temperature swings compared to inland areas.
-
Ocean Currents and Weather Patterns: Water's high heat capacity influences ocean currents, which distribute heat around the globe. This heat transfer influences weather patterns and climate systems worldwide.
Biological Significance:
-
Maintaining Body Temperature: The high heat capacity of water is essential for maintaining stable body temperatures in living organisms. Water makes up a significant portion of most organisms, acting as a thermal buffer that prevents rapid temperature changes.
-
Temperature Stability in Aquatic Ecosystems: Water's high heat capacity ensures relatively stable temperatures in aquatic ecosystems, providing a suitable habitat for a wide range of aquatic organisms.
Addressing the Misconception
The misconception that covalent bonds are responsible for water's low (it's actually high!) heat capacity arises from a misunderstanding of the different types of chemical bonds and their roles in determining a substance's properties. While the strength of the covalent bonds within a water molecule does contribute to its overall stability, it's the intermolecular forces, specifically the hydrogen bonds between water molecules, that primarily determine its high heat capacity. The energy needed to break these hydrogen bonds significantly impacts how much heat is required to raise water's temperature.
Conclusion: The Truth About Water's Heat Capacity
In conclusion, the assertion that covalent bonds give water a low heat capacity is fundamentally inaccurate. Water's remarkably high heat capacity is a direct consequence of the extensive network of hydrogen bonds formed between its molecules. These bonds require significant energy to break, leading to a substantial amount of heat absorption with only a small temperature increase. This unique property is crucial for regulating global climate patterns and supporting the diversity of life on Earth. Understanding the interplay between covalent and hydrogen bonds is key to grasping the full complexity and importance of water's extraordinary properties.
Latest Posts
Latest Posts
-
What Is 17 As A Percent
Apr 04, 2025
-
What Is A 15 Out Of 17
Apr 04, 2025
-
How Tall Is 73 Inches In Ft
Apr 04, 2025
-
How Many Meters In 4 Kilometers
Apr 04, 2025
-
Is Iodine A Metal Or Nonmetal
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Do Covalent Bonds Give Water A Low Heat Capacity . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
