Horizontal Rows On The Periodic Table Are Called
Kalali
Apr 05, 2025 · 7 min read
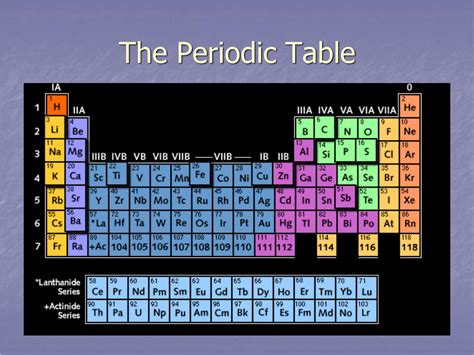
Table of Contents
Horizontal Rows on the Periodic Table are Called: Periods – A Deep Dive into Periodic Trends
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic number and recurring chemical properties. Understanding its structure is crucial for grasping the behavior of elements and predicting their reactions. A fundamental aspect of this organization involves the horizontal rows, which are known as periods. This article delves deep into the concept of periods, exploring their significance, the trends observed within them, and their importance in understanding the periodic law.
What are Periods in the Periodic Table?
Periods in the periodic table are the horizontal rows of elements arranged in order of increasing atomic number. Each period corresponds to a principal energy level (or shell) that is being filled with electrons. As we move across a period, the atomic number increases by one, meaning that one more proton and one more electron are added to each successive element. This addition of electrons influences the element's chemical properties, leading to observable trends.
Number of Periods and their Significance
The periodic table currently contains seven periods, each with a varying number of elements. The length of each period is determined by the number of electrons that can occupy the subshells within that principal energy level. This number follows the pattern: 2, 8, 8, 18, 18, 32, and (currently incomplete) 32.
- Period 1 (the shortest): Contains only two elements, hydrogen (H) and helium (He), as only the 1s subshell is filled in this period.
- Period 2 and 3: Both contain eight elements each, filling the 2s and 2p, and 3s and 3p subshells, respectively.
- Period 4 and 5: These periods have 18 elements each, due to the filling of the 3d subshell in addition to the 4s and 4p (period 4) and 5s and 5p (period 5) subshells. The 3d orbitals are filled after the 4s orbital, leading to the transition metals.
- Period 6 and 7: These periods have 32 elements, accommodating the filling of the 4f and 5d subshells, along with the 6s and 6p (period 6) and 7s and 7p (period 7). The 4f orbitals are filled after the 6s, leading to the lanthanides, and the 5f orbitals are filled after the 7s, leading to the actinides.
The varying lengths of periods directly reflect the complex electron configurations and the filling of different subshells within the atoms.
Periodic Trends within Periods
Moving across a period, we observe systematic changes in the properties of elements. These trends are largely due to the increase in the effective nuclear charge—the net positive charge experienced by the valence electrons—and the addition of electrons to the same principal energy level. Key trends include:
1. Atomic Radius: Decreasing Trend
Atomic radius, the distance from the nucleus to the outermost electron, generally decreases across a period. While the number of electrons increases, the added electrons are placed in the same energy level, closer to the nucleus. Simultaneously, the increasing number of protons increases the positive charge of the nucleus, pulling the electrons closer, thus reducing the atomic radius.
2. Ionization Energy: Increasing Trend
Ionization energy is the energy required to remove an electron from a gaseous atom. This value generally increases across a period. The increasing nuclear charge holds the electrons more tightly, requiring more energy to remove one.
3. Electronegativity: Increasing Trend
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. It generally increases across a period. As the nuclear charge increases and atomic radius decreases, the atom's ability to attract shared electrons in a bond increases.
4. Electron Affinity: Generally Increasing Trend
Electron affinity is the energy change when an electron is added to a neutral atom. While there are exceptions, generally electron affinity increases across a period due to the increasing nuclear charge pulling the added electron more strongly toward the nucleus.
5. Metallic Character: Decreasing Trend
Metallic character, the tendency of an element to lose electrons and form positive ions, generally decreases across a period. As we move from left to right, the elements become less likely to lose electrons because of the increasing nuclear charge and the increasing difficulty in removing electrons. The elements on the right side of the table are nonmetals, known for their tendency to gain electrons.
6. Chemical Reactivity: Variable Trend
Chemical reactivity is complex and depends on multiple factors. For example, alkali metals (Group 1) on the far left are highly reactive, readily losing one electron to achieve a stable electron configuration. Halogens (Group 17) on the right are also highly reactive, readily gaining one electron to achieve a stable octet. Noble gases (Group 18) are unreactive due to their stable electron configurations. The trend in reactivity is therefore not uniform across a period.
The Importance of Periods in Understanding the Periodic Law
The arrangement of elements in periods is fundamental to the periodic law, which states that the properties of elements are periodic functions of their atomic numbers. The repeating patterns of properties observed across periods are a direct consequence of the recurring electron configurations. The filling of electron shells and subshells leads to the observed trends in atomic radius, ionization energy, electronegativity, and other properties. This periodicity allows us to predict the properties of elements based on their position on the periodic table, even if they have not been fully characterized.
Beyond the Basic Trends: A Deeper Look at Periodicity
While the general trends described above provide a good overview, understanding the finer details requires considering other factors. For instance:
- Electron shielding: Inner electrons partially shield the outer electrons from the full effect of the nuclear charge. This shielding effect is not constant across a period and influences the observed trends.
- Subshell filling: The specific order of subshell filling (e.g., 4s before 3d) introduces variations in the trends. For example, the relatively low ionization energy of some transition metals is partly due to the shielding effect of the inner d-electrons.
- Anomalous behavior: There are exceptions to the general trends, particularly within transition metal series. This is attributed to the complex interplay of electron-electron repulsions and nuclear charge effects.
Applications of Periodicity: Predicting Chemical Behavior
The periodic table's organization by periods is vital for predicting the chemical behavior of elements. By understanding the trends associated with periods, chemists can:
- Predict the reactivity of elements: Knowledge of electronegativity and ionization energy allows us to anticipate how elements will behave in chemical reactions.
- Design new materials: The periodic table guides the selection of elements with specific properties for creating novel materials with desired characteristics (strength, conductivity, etc.).
- Understand chemical bonding: The positions of elements in a period help us understand the types of bonds (ionic, covalent, metallic) that they are likely to form.
- Interpret spectroscopic data: Trends in electronic configurations across periods can be correlated with spectroscopic data to help identify unknown elements or molecules.
Conclusion: Periods - The Foundation of Chemical Understanding
The horizontal rows, or periods, on the periodic table are not just an arbitrary arrangement. They are a fundamental aspect of the table's organization, reflecting the systematic filling of electron shells and subshells. Understanding the trends observed within periods is crucial for grasping the chemical behavior of elements and predicting their interactions. The periodic table, with its organized arrangement by periods, is a powerful tool that underpins much of our understanding of chemistry, impacting various scientific fields and technological advancements. The concept of periods, therefore, serves as a cornerstone of modern chemistry, allowing for predictions and interpretations that drive scientific progress. From the simplest reactions to the creation of complex materials, the influence of period trends is undeniable. The periodic table’s structure, including its periods, stands as a testament to the elegance and power of scientific organization, enabling a deeper comprehension of the matter that surrounds us.
Latest Posts
Latest Posts
-
1 6 M In Feet And Inches
Apr 05, 2025
-
How Many Inches Is 124 Cm
Apr 05, 2025
-
Can A Sedimentary Rock Become An Igneous Rock
Apr 05, 2025
-
How Many Feet Is 104 In
Apr 05, 2025
-
How Tall Is 54 In In Feet
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Horizontal Rows On The Periodic Table Are Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
