How To Find The Percent Recovery
Kalali
Apr 03, 2025 · 6 min read
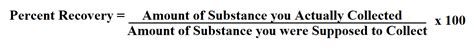
Table of Contents
How to Find Percent Recovery: A Comprehensive Guide
Percent recovery, a crucial concept in various scientific fields and industrial processes, represents the efficiency of a process in retrieving a desired substance or product. Understanding how to calculate and interpret percent recovery is essential for optimizing procedures, minimizing waste, and ensuring accurate results. This comprehensive guide delves into the intricacies of percent recovery, providing you with a step-by-step approach, practical examples, and insights into its broader implications.
Understanding the Concept of Percent Recovery
Percent recovery quantifies the amount of a target substance successfully retrieved relative to the initial amount present. It's expressed as a percentage, indicating the effectiveness of a separation or purification technique. A high percent recovery signifies a highly efficient process, while a low percentage points to potential losses or inefficiencies that need addressing. This concept finds application in diverse areas, including:
- Chemistry: Isolating and purifying compounds from mixtures or reactions.
- Environmental Science: Analyzing the concentration of pollutants in samples.
- Pharmaceuticals: Assessing the yield of drug synthesis.
- Mining and Metallurgy: Determining the efficiency of extracting valuable metals from ores.
- Food Science: Measuring the yield of processed food products.
Calculating Percent Recovery: A Step-by-Step Guide
The fundamental formula for calculating percent recovery is straightforward:
Percent Recovery = [(Actual Yield / Theoretical Yield) x 100]%
Let's break down each component:
1. Determining the Theoretical Yield
The theoretical yield represents the maximum amount of product that could be obtained under ideal conditions. This value is calculated based on stoichiometry, the quantitative relationships between reactants and products in a chemical reaction. It's crucial to have a balanced chemical equation to accurately predict the theoretical yield.
Example: Consider a reaction where 10 grams of reactant A completely reacts to produce product B. If the molar mass of A is 50 g/mol and the molar mass of B is 100 g/mol, and the reaction stoichiometry is 1:1 (1 mole of A produces 1 mole of B), the theoretical yield calculation would be:
- Moles of A = (10 g) / (50 g/mol) = 0.2 mol
- Since the stoichiometry is 1:1, 0.2 mol of B is produced.
- Mass of B (Theoretical Yield) = (0.2 mol) x (100 g/mol) = 20 g
Therefore, the theoretical yield of product B is 20 grams.
2. Determining the Actual Yield
The actual yield is the actual amount of product obtained after conducting the experiment or process. This value is determined through experimental measurements, such as weighing the isolated product or using other appropriate analytical techniques. It's crucial to account for any losses during the process (e.g., material sticking to glassware, incomplete reactions, filtration losses).
Example (Continuing from above): Let's say after conducting the experiment, you isolate and weigh 18 grams of product B. This is your actual yield.
3. Calculating Percent Recovery
Now, we can plug the actual and theoretical yields into the formula:
Percent Recovery = [(18 g / 20 g) x 100]% = 90%
This means the process had a 90% recovery rate. 10% of the product was lost during the procedure.
Factors Affecting Percent Recovery
Several factors can influence the percent recovery obtained in a process:
1. Incomplete Reactions:
Reactions may not proceed to completion, resulting in a lower actual yield than theoretically predicted. Factors like reaction kinetics, equilibrium limitations, and insufficient reaction time contribute to incomplete reactions.
2. Loss of Product During Purification:
Purification steps like filtration, crystallization, or extraction can lead to product loss. Some product might remain adsorbed onto filter paper, lost during transfer between containers, or remain in the mother liquor during crystallization.
3. Side Reactions:
Unwanted side reactions can consume reactants, reducing the amount available for the desired product formation. These side reactions often generate byproducts, complicating the purification process and lowering recovery.
4. Experimental Errors:
Errors in measurements, sample handling, or equipment malfunction can affect the accuracy of both actual and theoretical yield calculations, impacting the percent recovery.
5. Purity of Reactants:
Impurities in the starting reactants can reduce the actual yield, either by interfering with the main reaction or by introducing side reactions.
6. Environmental Conditions:
Temperature, pressure, and solvent choice can significantly influence reaction yield and consequently, percent recovery.
Improving Percent Recovery
Strategies for improving percent recovery involve addressing the factors mentioned above:
-
Optimizing Reaction Conditions: Fine-tuning parameters like temperature, pressure, reaction time, and the use of catalysts can enhance reaction efficiency and minimize side reactions.
-
Improving Purification Techniques: Employing more efficient separation techniques, using appropriate solvents, and minimizing transfers can reduce product loss.
-
Careful Handling and Measurement: Minimizing sample handling errors, using accurate measuring instruments, and performing multiple trials can improve the accuracy of results.
-
Using High-Purity Reactants: Ensuring the reactants are pure minimizes the impact of impurities on reaction yield.
-
Analyzing Losses: Identifying the specific stages where losses occur allows for targeted improvements in the process. Detailed analysis might reveal that improvements are needed in a particular purification stage.
Interpreting Percent Recovery Results
The percent recovery value provides insights into the efficiency of the process. A high percentage, typically above 90%, generally indicates a well-optimized and efficient process. However, the acceptable range for percent recovery can vary widely based on the nature of the process, the desired purity level, and the complexity of the procedure. Low recovery requires critical evaluation to identify the causes and implement improvements.
Beyond the Basics: Advanced Applications
The concept of percent recovery extends beyond simple chemical reactions. In fields like environmental science, percent recovery is used to assess the efficiency of pollutant removal from contaminated sites. In medicine, it can help assess the effectiveness of drug delivery systems. In material science, the percent recovery of a specific material from a composite is crucial for characterizing the material's properties.
The methodology might vary slightly depending on the specific application. For example, in environmental science, the theoretical yield could be the total amount of pollutant initially present in a sample, while the actual yield is the amount recovered after a remediation process.
Conclusion
Understanding how to calculate and interpret percent recovery is a fundamental skill for anyone involved in chemistry, engineering, environmental science, or any field involving the isolation and purification of substances. By systematically analyzing the factors that influence percent recovery and employing appropriate techniques to optimize the process, significant improvements in efficiency and accuracy can be achieved. This detailed guide serves as a comprehensive resource for mastering this essential concept, helping you to improve your experimental design, data analysis, and overall understanding of chemical processes. Remember that attention to detail, proper experimental technique, and a thorough understanding of the underlying principles are key to achieving high percent recovery.
Latest Posts
Latest Posts
-
What Percent Of 400 Is 20
Apr 04, 2025
-
How Many Sides Does A Trapezoid Has
Apr 04, 2025
-
Antiderivative Of The Square Root Of X
Apr 04, 2025
-
Lowest Common Multiple Of 24 And 16
Apr 04, 2025
-
Is Aluminium A Metal Nonmetal Or Metalloid
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about How To Find The Percent Recovery . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
