Is Non Polar Hydrophobic Or Hydrophilic
Kalali
Apr 01, 2025 · 5 min read
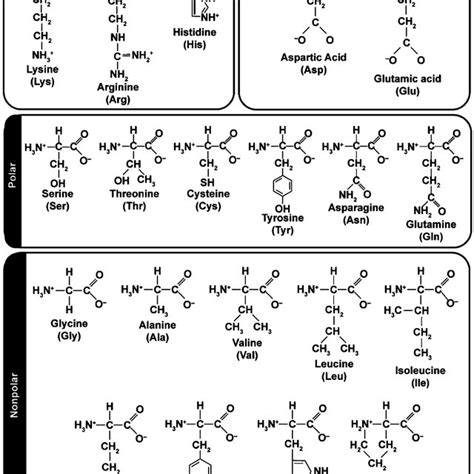
Table of Contents
- Is Non Polar Hydrophobic Or Hydrophilic
- Table of Contents
- Is Nonpolar Hydrophobic or Hydrophilic? Understanding Polarity and Molecular Interactions
- Polarity: The Foundation of Molecular Interactions
- Hydrophobicity vs. Hydrophilicity: Attraction and Repulsion
- The Link Between Nonpolarity and Hydrophobicity: Why Nonpolar Molecules are Hydrophobic
- Exceptions and Nuances: Not all Nonpolar Molecules are Equally Hydrophobic
- Measuring Hydrophobicity: Quantitative Approaches
- Applications of Understanding Hydrophobicity: From Biology to Materials Science
- Conclusion: Hydrophobicity is a Complex Phenomenon
- Latest Posts
- Latest Posts
- Related Post
Is Nonpolar Hydrophobic or Hydrophilic? Understanding Polarity and Molecular Interactions
The question of whether nonpolar molecules are hydrophobic or hydrophilic is fundamental to understanding chemistry and biology. The terms often get used interchangeably, leading to confusion. However, a clear distinction exists, and understanding this difference is crucial for comprehending how molecules interact and behave in various environments. This comprehensive article will delve into the intricacies of polarity, hydrophobicity, and hydrophilicity, clarifying the relationship between nonpolarity and hydrophobicity.
Polarity: The Foundation of Molecular Interactions
Before diving into hydrophobicity and hydrophilicity, we must first grasp the concept of polarity. Polarity describes the distribution of electrical charge within a molecule. Molecules are composed of atoms bonded together, and these atoms have different electronegativities – a measure of their ability to attract electrons in a chemical bond.
-
Polar molecules: In polar molecules, there's an uneven distribution of charge. One end of the molecule carries a partial positive charge (δ+), while the other end carries a partial negative charge (δ−). This is typically due to the presence of polar covalent bonds, where electrons are shared unequally between atoms of differing electronegativities. Water (H₂O) is a classic example of a polar molecule. The oxygen atom is more electronegative than the hydrogen atoms, pulling the electrons closer and creating a partial negative charge on the oxygen and partial positive charges on the hydrogens.
-
Nonpolar molecules: In nonpolar molecules, the electrons are shared relatively equally between atoms. This results in a balanced charge distribution, with no significant partial charges. Many organic molecules, such as hydrocarbons (molecules containing only carbon and hydrogen), are nonpolar. Examples include methane (CH₄) and hexane (C₆H₁₄).
Hydrophobicity vs. Hydrophilicity: Attraction and Repulsion
Now, let's examine the terms hydrophobicity and hydrophilicity. These terms describe how molecules interact with water, which is a highly polar solvent.
-
Hydrophilic: Hydrophilic molecules are "water-loving." They readily interact with and dissolve in water. This interaction is driven by the attraction between the polar molecules and the polar water molecules. Polar molecules can form hydrogen bonds with water, contributing to their solubility. Examples of hydrophilic molecules include sugars, alcohols, and many salts.
-
Hydrophobic: Hydrophobic molecules are "water-fearing." They tend to avoid contact with water and do not readily dissolve in it. This aversion stems from the fact that nonpolar molecules cannot form hydrogen bonds or strong dipole-dipole interactions with water molecules. Instead, they disrupt the highly organized hydrogen bonding network of water, leading to a thermodynamically unfavorable state. This is why hydrophobic molecules tend to cluster together to minimize their contact with water. Examples of hydrophobic molecules include fats, oils, and many hydrocarbons.
The Link Between Nonpolarity and Hydrophobicity: Why Nonpolar Molecules are Hydrophobic
The crucial connection is that most nonpolar molecules are hydrophobic. This is because their inability to form strong interactions with water molecules leads to their repulsion by water. The water molecules, in an attempt to maintain their highly ordered hydrogen bonding network, effectively "push out" the nonpolar molecules.
This hydrophobic effect is a major driving force in many biological processes, including:
-
Protein folding: Hydrophobic amino acid side chains tend to cluster in the protein's interior, away from the aqueous environment, while hydrophilic side chains are typically located on the protein's surface.
-
Membrane formation: Cell membranes are composed of a lipid bilayer, where the hydrophobic tails of the phospholipids face inwards, away from the water, while the hydrophilic heads face outwards, interacting with the surrounding aqueous environment.
-
Micelle formation: Amphiphilic molecules (molecules with both hydrophilic and hydrophobic regions), such as surfactants, spontaneously form micelles in water. The hydrophobic tails cluster together in the interior of the micelle, minimizing their contact with water, while the hydrophilic heads face outwards, interacting with the water.
Exceptions and Nuances: Not all Nonpolar Molecules are Equally Hydrophobic
While the connection between nonpolarity and hydrophobicity is strong, it's not absolute. Some nonpolar molecules exhibit weaker hydrophobic interactions than others. The size and shape of the molecule, as well as its ability to induce dipole moments in water molecules, can influence its hydrophobicity.
Furthermore, the term "hydrophobic" doesn't imply a complete absence of interaction with water. Instead, it describes a weaker preference for interacting with water compared to other molecules or itself. There can be weak van der Waals forces between nonpolar molecules and water, but these are significantly weaker than the hydrogen bonds between water molecules.
Measuring Hydrophobicity: Quantitative Approaches
The hydrophobicity of molecules can be quantified using various methods, such as:
-
Partition coefficients: These measure the relative solubility of a molecule in two immiscible solvents, typically water and octanol. A higher partition coefficient indicates greater hydrophobicity.
-
Hydrophobicity scales: These assign numerical values to the hydrophobicity of different amino acids or other molecules, based on experimental data or theoretical calculations. These scales are often used in predicting protein structure and function.
-
Contact angle measurements: This method measures the angle formed by a droplet of water on a surface. A higher contact angle indicates greater hydrophobicity of the surface.
Applications of Understanding Hydrophobicity: From Biology to Materials Science
Understanding the principles of hydrophobicity and hydrophilicity is essential in numerous fields:
-
Drug delivery: Hydrophobicity plays a crucial role in designing drug formulations, influencing how drugs are absorbed and distributed in the body.
-
Materials science: Creating hydrophobic or hydrophilic materials with specific properties is vital in various applications, from creating waterproof fabrics to designing self-cleaning surfaces.
-
Environmental science: Understanding how hydrophobic pollutants behave in the environment is crucial for developing effective remediation strategies.
-
Biotechnology: Hydrophobicity is fundamental to many biotechnological processes, such as protein purification and enzyme immobilization.
Conclusion: Hydrophobicity is a Complex Phenomenon
In summary, while nonpolar molecules are generally hydrophobic, it's crucial to remember that hydrophobicity is a complex phenomenon influenced by various factors beyond simple polarity. It's not an absolute property but rather a relative measure of a molecule's interaction with water, driven by the thermodynamic preference for maximizing water's hydrogen bonding network. Understanding this delicate balance between polar and nonpolar interactions is paramount in numerous scientific disciplines. Further research continues to refine our understanding of these complex molecular interactions, opening doors to innovative applications in diverse fields. The interplay between polarity, hydrophobicity, and hydrophilicity remains a cornerstone of scientific inquiry, leading to breakthroughs in fields ranging from medicine and materials science to environmental protection and beyond.
Latest Posts
Latest Posts
-
8 Of 15 Is What Percentage
Apr 05, 2025
-
60 Ounces Is How Many Liters
Apr 05, 2025
-
A Single Replacement Reaction Occurs When
Apr 05, 2025
-
How Big Is 14 Cm In Inches
Apr 05, 2025
-
How Many Liters Are In 60 Ounces
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Is Non Polar Hydrophobic Or Hydrophilic . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
