Magnesium Metal Or Nonmetal Or Metalloid
Kalali
Apr 02, 2025 · 6 min read
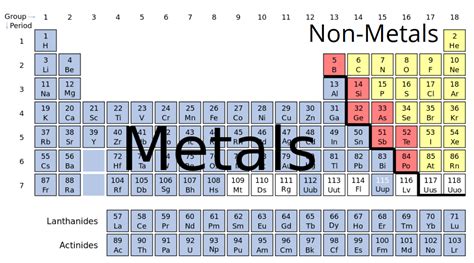
Table of Contents
Magnesium: A Deep Dive into the Reactive Alkaline Earth Metal
Magnesium, a silvery-white metal, occupies a prominent position in the periodic table, nestled within Group 2, the alkaline earth metals. While often overlooked in everyday conversations, magnesium plays a crucial role in numerous industrial processes and biological functions. Understanding its properties, applications, and importance is essential for appreciating its multifaceted nature. This comprehensive article will explore magnesium's classification, characteristics, production, applications, and biological significance.
Is Magnesium a Metal, Nonmetal, or Metalloid?
The question of whether magnesium is a metal, nonmetal, or metalloid is easily answered: magnesium is a metal. Its properties firmly place it within the metallic category. Metalloids exhibit characteristics of both metals and nonmetals, sitting on the border between the two groups. Nonmetals, on the other hand, lack the characteristic properties of metals such as conductivity and malleability. Magnesium, however, clearly displays metallic traits.
Key Metallic Properties of Magnesium:
- Excellent Electrical and Thermal Conductivity: Magnesium readily conducts electricity and heat, a defining feature of metals.
- Malleability and Ductility: It can be easily shaped and drawn into wires, demonstrating malleability and ductility.
- Metallic Luster: Magnesium possesses a characteristic metallic shine.
- Low Ionization Energy: It readily loses electrons to form positive ions (Mg²⁺), a common trait among metals.
The Abundant Alkaline Earth Metal: Occurrence and Production
Magnesium is surprisingly abundant in the Earth's crust, ranking as the eighth most abundant element. However, it is rarely found in its pure metallic form due to its high reactivity. It primarily occurs in combination with other elements, most notably in minerals like:
- Dolomite (CaMg(CO₃)₂): A carbonate mineral, often found in large sedimentary deposits.
- Magnesite (MgCO₃): Another carbonate mineral, a valuable source of magnesium.
- Carnallite (KCl·MgCl₂·6H₂O): An evaporite mineral containing magnesium chloride.
- Seawater: Seawater contains a significant amount of magnesium ions, representing a vast, albeit dilute, resource.
Extraction and Refining of Magnesium:
Several methods are employed for extracting magnesium from its ores and seawater:
- Electrolysis of Molten Magnesium Chloride (MgCl₂): This is the most common method, where molten magnesium chloride is electrolyzed to yield pure magnesium metal. The process requires high temperatures to melt the magnesium chloride and significant energy input.
- Pidgeon Process: This method utilizes the reduction of magnesium oxide (MgO) with ferrosilicon (an alloy of iron and silicon) under vacuum at high temperatures. It produces relatively pure magnesium, although it is less efficient than electrolysis.
- Extraction from Seawater: Seawater contains magnesium ions (Mg²⁺). A chemical process involving the precipitation of magnesium hydroxide (Mg(OH)₂), followed by conversion to magnesium chloride, and finally electrolysis, is used to extract magnesium from seawater. This method is becoming increasingly important due to the vast and readily available resource.
Diverse Applications of Magnesium: A Versatile Metal
Magnesium's unique combination of properties—light weight, high strength-to-weight ratio, excellent machinability, and biocompatibility—has led to a wide range of applications across various industries.
Lightweight Alloys in Transportation:
Magnesium's low density (approximately 1.74 g/cm³) makes it an ideal material for lightweight alloys used in the automotive, aerospace, and transportation industries. These alloys contribute to improved fuel efficiency, reduced emissions, and enhanced vehicle performance. Examples include:
- Automotive parts: Wheels, engine blocks, transmission cases, and other components.
- Aerospace components: Aircraft parts, satellite structures, and rocket components.
- Sporting goods: Bicycle frames, golf clubs, and other sporting equipment.
Electronics and Electrical Applications:
Magnesium's electrical conductivity and corrosion resistance find applications in various electronic components:
- Battery anodes: Magnesium is explored as a potential anode material in next-generation batteries due to its high energy density.
- Semiconductors: Magnesium is used in some semiconductor manufacturing processes.
- Electronic shielding: It can be used for shielding sensitive electronic components from electromagnetic interference.
Biomedical Applications:
Magnesium's biocompatibility and biodegradable nature make it suitable for several biomedical applications:
- Biodegradable implants: Magnesium alloys are being developed as biodegradable implants, offering a less invasive alternative to traditional metallic implants. These implants gradually degrade within the body, eliminating the need for a secondary surgery for removal.
- Drug delivery systems: Magnesium can be incorporated into drug delivery systems for controlled release of pharmaceuticals.
- Dental applications: It finds uses in certain dental materials.
Other Notable Applications:
- Pyrotechnics: Magnesium burns with a bright white light, making it a key component in flares, fireworks, and incendiary devices.
- Metallurgy: It's used as an alloying agent in aluminum and other metal alloys to enhance their properties.
- Chemical Industry: Magnesium is used as a reducing agent in various chemical reactions.
Biological Significance of Magnesium: An Essential Element
Beyond its industrial applications, magnesium plays a vital role in biological systems. It is an essential mineral for humans and other living organisms, participating in numerous metabolic processes.
Magnesium's Role in Biological Processes:
- Enzyme Activation: Magnesium acts as a cofactor for many enzymes involved in crucial metabolic reactions, including those related to carbohydrate metabolism, protein synthesis, and DNA replication.
- Muscle and Nerve Function: It is essential for muscle contraction and nerve impulse transmission. Magnesium deficiency can lead to muscle cramps, tremors, and neurological problems.
- Bone Health: Magnesium contributes to bone mineralization and strength. Adequate magnesium intake is crucial for maintaining strong and healthy bones.
- Blood Pressure Regulation: It helps regulate blood pressure.
- Blood Sugar Control: Magnesium plays a role in maintaining healthy blood sugar levels.
- Protein Synthesis: Magnesium is crucial in the synthesis of proteins.
Magnesium Deficiency and its Consequences:
Magnesium deficiency, or hypomagnesemia, can result from various factors, including inadequate dietary intake, malabsorption, excessive alcohol consumption, and certain medical conditions. Symptoms of magnesium deficiency can include muscle weakness, fatigue, tremors, nausea, vomiting, and personality changes. Severe deficiency can lead to more serious complications, such as seizures and cardiac arrhythmias.
Dietary Sources of Magnesium:
Maintaining adequate magnesium levels through diet is crucial. Good dietary sources of magnesium include:
- Leafy green vegetables: Spinach, kale, and collard greens.
- Legumes: Beans, lentils, and peas.
- Nuts and seeds: Almonds, cashews, pumpkin seeds, and sunflower seeds.
- Whole grains: Oats, brown rice, and quinoa.
- Dark chocolate: In moderation.
Environmental Considerations and Sustainability:
While magnesium is a relatively abundant element, its extraction and processing require significant energy consumption and can generate environmental concerns. The electrolysis process, for instance, is energy-intensive. Furthermore, the disposal of magnesium-containing waste materials needs careful consideration. Sustainable practices in magnesium production and recycling are essential to minimize its environmental footprint.
Future Research and Development:
Research continues to explore new and sustainable methods for magnesium extraction and processing, focusing on reducing energy consumption and minimizing environmental impact. The development of more efficient magnesium alloys and improved recycling techniques are also areas of ongoing research.
Conclusion: A Metal of Multifaceted Importance
Magnesium, a versatile and abundant alkaline earth metal, holds a pivotal position in various industrial processes and biological systems. Its unique properties, ranging from its lightweight nature to its biocompatibility, have led to a remarkable diversity of applications. From lightweight alloys in transportation to essential roles in biological processes, magnesium's importance is undeniable. However, responsible and sustainable practices in its extraction, processing, and disposal are vital to ensure its continued utilization while mitigating potential environmental impacts. As research continues to unveil new applications and refine existing methods, magnesium's influence across various sectors is poised to further expand. The journey of magnesium, from its abundance in the Earth's crust to its myriad applications and biological significance, underlines its remarkable and multifaceted nature, showcasing it as a truly vital element in both the industrial and biological realms.
Latest Posts
Latest Posts
-
How Tall Is 35 Inches In Feet
Apr 03, 2025
-
44 Is What Percent Of 50
Apr 03, 2025
-
36 Feet Is How Many Meters
Apr 03, 2025
-
What Is 8 12 As A Percent
Apr 03, 2025
-
1 73 M In Inches And Feet
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Magnesium Metal Or Nonmetal Or Metalloid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
