Rutherfords Gold Foil Experiment Determined That
Kalali
Apr 02, 2025 · 6 min read
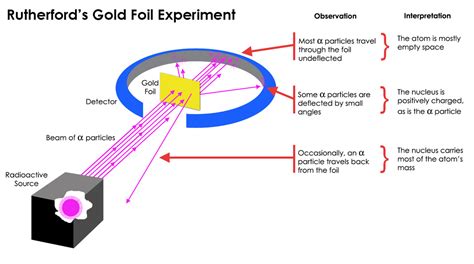
Table of Contents
Rutherford's Gold Foil Experiment: Determining the Structure of the Atom
Rutherford's gold foil experiment, conducted in 1909 by Hans Geiger and Ernest Marsden under the supervision of Ernest Rutherford, revolutionized our understanding of the atom. Before this groundbreaking experiment, the prevailing model of the atom was the "plum pudding" model proposed by J.J. Thomson. This model depicted the atom as a positively charged sphere with negatively charged electrons embedded within it, like plums in a pudding. Rutherford's experiment, however, shattered this model and paved the way for the modern understanding of the atom's structure, revealing the existence of a tiny, dense, positively charged nucleus at the center.
The Experiment: A Simple Setup with Profound Implications
The experimental setup was deceptively simple. A beam of alpha particles (positively charged particles emitted by radioactive substances) was directed at a very thin gold foil (only a few atoms thick). Surrounding the gold foil was a fluorescent screen that would detect the scattered alpha particles. The expectation, based on Thomson's model, was that the alpha particles would pass straight through the gold foil with only minor deflections. After all, if the positive charge was uniformly distributed throughout the atom, there shouldn't be any significant forces to deflect the positively charged alpha particles.
Unexpected Results: A Paradigm Shift
The results, however, were astonishing. While most of the alpha particles did pass straight through the foil as expected, a significant number were deflected at large angles, and some even bounced straight back! This was completely unexpected and directly contradicted the predictions of the plum pudding model. It was as if the alpha particles were encountering a small, concentrated region of intense positive charge within the atom.
Interpreting the Results: The Birth of the Nuclear Model
Rutherford meticulously analyzed the scattering data. The vast majority of alpha particles passing through the foil undisturbed indicated that most of the atom is empty space. However, the significant number of deflections, especially the backscattering, suggested the presence of a dense, positively charged core within the atom. This core, he reasoned, must be responsible for the strong repulsive forces that caused the deflections.
The Nuclear Model: A Revolutionary Concept
Based on these observations, Rutherford proposed the nuclear model of the atom. This model posited that:
- The atom is mostly empty space: The fact that most alpha particles passed straight through indicated that the atom consists primarily of empty space.
- A small, dense, positively charged nucleus exists at the center: This nucleus contains most of the atom's mass and all of its positive charge.
- Electrons orbit the nucleus: Negatively charged electrons orbit the nucleus at a relatively large distance, balancing the positive charge of the nucleus.
This model dramatically differed from Thomson's plum pudding model. Instead of a uniform distribution of charge, Rutherford's model depicted a highly concentrated positive charge in the nucleus, surrounded by a vast expanse of empty space containing orbiting electrons.
Significance of Rutherford's Gold Foil Experiment
Rutherford's gold foil experiment was a landmark achievement in the history of physics. Its impact extends far beyond simply revealing the structure of the atom. Here are some of its key significances:
-
Foundation of modern atomic theory: The experiment provided the foundation for the modern understanding of the atom's structure, which continues to be refined and expanded upon today. The concept of a nucleus surrounded by orbiting electrons is fundamental to our understanding of chemistry, physics, and many other scientific disciplines.
-
Development of nuclear physics: The discovery of the nucleus directly led to the development of nuclear physics, a branch of physics that studies the structure and properties of atomic nuclei. This field has had a profound impact on our world, leading to advancements such as nuclear energy and nuclear medicine.
-
Improved understanding of radioactive decay: The use of alpha particles in the experiment further advanced the understanding of radioactive decay and the properties of radioactive materials. This understanding has had significant implications for various fields, including medicine, environmental science, and archaeology.
-
Advancements in experimental techniques: The experiment itself demonstrated the power of experimental techniques to reveal fundamental truths about the natural world. The meticulous planning, execution, and interpretation of the data established a new standard for scientific investigation. The experiment showcased the importance of carefully designed experiments in probing the fundamental structures of matter.
-
Paradigm shift in scientific thinking: The results of the gold foil experiment forced a fundamental shift in scientific thinking about the nature of matter. It showed that the seemingly simple plum pudding model was incorrect and that a more sophisticated and complex model was needed to explain the experimental observations. This highlighted the crucial role of experimental evidence in validating or refuting scientific theories.
Limitations and Further Developments
While revolutionary, Rutherford's model had its limitations. It couldn't explain certain aspects of atomic behavior, such as the stability of atoms and the discrete nature of atomic spectra (the specific wavelengths of light emitted by atoms). These limitations were addressed by later models, most notably Bohr's model, which incorporated the concept of quantized energy levels for electrons. Bohr's model, while an improvement, still had limitations and eventually gave way to the more sophisticated quantum mechanical model of the atom, which provides the most accurate description of atomic structure and behavior to date. However, Rutherford's experiment laid the crucial groundwork for these later developments.
The Lasting Legacy
Rutherford's gold foil experiment remains a cornerstone of modern physics education. It’s a powerful example of how a seemingly simple experiment can lead to revolutionary discoveries and a profound shift in our understanding of the universe. The experiment's simplicity and the dramatic contrast between expectation and results make it a compelling case study in scientific inquiry. It teaches us the importance of meticulous observation, rigorous analysis, and the willingness to question established models in the pursuit of scientific truth. The experiment’s enduring legacy lies not just in its specific findings but also in its exemplary demonstration of the scientific method in action, inspiring generations of scientists and students alike.
Beyond the Basics: Exploring Further Research
The gold foil experiment opened up numerous avenues for further research. Scientists built upon Rutherford’s findings to investigate:
-
Nuclear structure: The nature of the nucleus itself became a major area of research, leading to discoveries about protons, neutrons, and the strong nuclear force that holds them together.
-
Atomic spectra: The discrepancies between Rutherford’s model and observed atomic spectra led to the development of quantum mechanics and a deeper understanding of electron behavior.
-
Nuclear reactions: The knowledge gained from understanding the nucleus paved the way for the study of nuclear reactions, leading to technologies like nuclear power and nuclear medicine.
-
Particle physics: The experiment provided foundational knowledge for the development of particle physics, which explores the fundamental building blocks of matter.
The impact of Rutherford's work extends far beyond the specific details of atomic structure. His experimental approach and the resulting paradigm shift in scientific understanding represent a triumph of the scientific method and continue to inspire scientific inquiry today. The simple gold foil experiment stands as a testament to the power of observation, ingenuity, and the relentless pursuit of knowledge. Its legacy continues to resonate within the scientific community, reminding us of the profound impact of a single, meticulously executed experiment.
Latest Posts
Latest Posts
-
Find All Complex Zeros Of The Polynomial Function
Apr 03, 2025
-
How Many Cups To 20 Oz
Apr 03, 2025
-
What Is 230 Degrees Celsius In Fahrenheit
Apr 03, 2025
-
Cuanto Es El 25 De 100
Apr 03, 2025
-
How Many Gallons Are In 3 Liters
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Rutherfords Gold Foil Experiment Determined That . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
