What Is A Property Of A Base
Kalali
Apr 05, 2025 · 7 min read
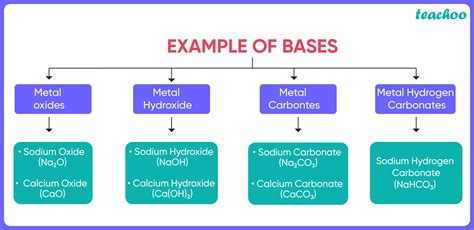
Table of Contents
What is a Property of a Base? A Deep Dive into Base Characteristics
Understanding the properties of bases is fundamental to chemistry. Bases, alongside acids, form the cornerstone of acid-base chemistry, a crucial concept in numerous fields, from environmental science to medicine. This comprehensive guide explores the defining characteristics of bases, explaining their behavior, reactions, and significance in various applications. We'll delve into both theoretical and practical aspects, clarifying common misconceptions and providing a robust understanding of this essential chemical concept.
Defining Bases: Arrhenius, Brønsted-Lowry, and Lewis Theories
The definition of a base has evolved over time, with different theories offering increasingly comprehensive perspectives.
Arrhenius Definition: The Hydroxide Ion Connection
The simplest definition, proposed by Svante Arrhenius, defines a base as a substance that dissociates in water to produce hydroxide ions (OH⁻). This definition is limited as it only applies to aqueous solutions. Strong Arrhenius bases, like sodium hydroxide (NaOH) and potassium hydroxide (KOH), completely dissociate, releasing a high concentration of OH⁻ ions. Weak Arrhenius bases, such as ammonia (NH₃), partially dissociate, resulting in a lower concentration of OH⁻ ions. This difference influences their pH and reactivity.
Brønsted-Lowry Definition: Proton Acceptors
The Brønsted-Lowry theory expands the definition by focusing on proton (H⁺) transfer. A Brønsted-Lowry base is defined as a proton acceptor. This definition encompasses a broader range of substances than the Arrhenius definition, including those that don't contain hydroxide ions but can still accept protons. For example, ammonia (NH₃) acts as a Brønsted-Lowry base by accepting a proton from water to form the ammonium ion (NH₄⁺). This theory is more versatile because it can describe acid-base reactions in non-aqueous solvents.
Lewis Definition: Electron Pair Donors
The most general definition is provided by the Lewis theory. A Lewis base is defined as an electron pair donor. This broadens the scope even further, including molecules that don't necessarily contain hydroxide ions or accept protons but possess a lone pair of electrons available for donation to an electron-deficient species (a Lewis acid). This definition is particularly useful in understanding reactions in organic chemistry and coordination chemistry. Many molecules that aren't considered bases according to the Arrhenius or Brønsted-Lowry definitions can act as Lewis bases. For example, ammonia (NH₃) has a lone pair of electrons on the nitrogen atom and can act as a Lewis base by donating this electron pair to a Lewis acid.
Key Properties of Bases
Bases exhibit several characteristic properties that help in their identification and understanding. These properties stem from their fundamental ability to accept protons or donate electron pairs.
1. pH Greater Than 7
One of the most readily measurable properties of a base is its pH. On the pH scale (ranging from 0 to 14), a pH greater than 7 indicates a basic solution. The higher the pH, the stronger the base. This is due to the increased concentration of hydroxide ions (OH⁻) in solution. Strong bases have a pH close to 14, while weak bases have a pH closer to 7.
2. Bitter Taste and Slippery Feel
While caution should always be exercised before tasting chemicals, bases typically possess a bitter taste and a slippery or soapy feel when dissolved in water. This slippery feel is caused by the reaction of the base with the oils and proteins on the skin, producing soap-like substances.
3. Reaction with Acids (Neutralization)
Bases readily react with acids in a process called neutralization. This reaction involves the combination of H⁺ ions from the acid and OH⁻ ions from the base to form water (H₂O). The resulting solution is usually less acidic or basic than the original solutions, depending on the relative strengths of the acid and base. The neutralization reaction often produces a salt, which is an ionic compound composed of the cation from the base and the anion from the acid.
4. Indicator Color Changes
Certain substances, known as indicators, change color in the presence of acids or bases. These indicators can be used to determine whether a solution is acidic, neutral, or basic. For instance, litmus paper turns blue in a basic solution and red in an acidic solution. Other indicators, such as phenolphthalein, undergo more dramatic color changes depending on the pH range.
5. Electrical Conductivity
Many bases, especially strong bases, are good conductors of electricity when dissolved in water. This is because the dissociation of the base produces ions (cations and anions), which are responsible for carrying electric current. Weak bases, however, show lower conductivity due to their limited dissociation.
6. Reactivity with Metals
Some bases, particularly those containing highly reactive metals, can react with certain metals, particularly amphoteric metals like aluminum and zinc, producing hydrogen gas (H₂). This reaction is a redox reaction where the metal is oxidized and the water (often present in the basic solution) is reduced.
Strong vs. Weak Bases: A Comparative Analysis
The strength of a base refers to its ability to dissociate in water and produce hydroxide ions.
Strong Bases
Strong bases completely dissociate in water, resulting in a high concentration of hydroxide ions. Examples include:
- Group 1 hydroxides: NaOH (sodium hydroxide), KOH (potassium hydroxide), LiOH (lithium hydroxide)
- Group 2 hydroxides: Ca(OH)₂ (calcium hydroxide), Ba(OH)₂ (barium hydroxide), Sr(OH)₂ (strontium hydroxide)
These bases readily donate their hydroxide ions and have high pH values.
Weak Bases
Weak bases partially dissociate in water, producing a relatively low concentration of hydroxide ions. Examples include:
- Ammonia (NH₃): Reacts with water to form a small amount of ammonium (NH₄⁺) and hydroxide (OH⁻) ions.
- Many organic amines: Molecules containing nitrogen with a lone pair of electrons.
Weak bases have lower pH values compared to strong bases. Their equilibrium lies significantly to the left, meaning most of the base remains undissociated.
Applications of Bases
Bases are widely used in numerous applications across various industries:
1. Industrial Applications
- Manufacturing of soaps and detergents: Sodium hydroxide (NaOH) is a key component in the saponification process, which involves the reaction of fats and oils with a strong base to produce soap.
- Pulp and paper industry: Bases are used in the pulping process to separate lignin from cellulose fibers.
- Chemical synthesis: Many chemical reactions utilize bases as catalysts or reagents.
2. Everyday Applications
- Antacids: Many antacids contain bases, such as calcium carbonate (CaCO₃) or magnesium hydroxide (Mg(OH)₂), to neutralize excess stomach acid.
- Cleaning agents: Many household cleaners are basic solutions, effectively removing grease and grime.
- Food industry: Bases are used in food processing, for example, to adjust pH in baking and to neutralize acidic compounds.
3. Environmental Applications
- Water treatment: Bases are used to adjust the pH of water to make it suitable for drinking and other purposes.
- Wastewater treatment: Bases are employed to neutralize acidic waste streams.
Safety Precautions When Handling Bases
Bases can be corrosive and harmful; therefore, proper safety precautions should always be followed:
- Wear appropriate personal protective equipment (PPE): This includes gloves, eye protection, and lab coats.
- Handle bases carefully: Avoid direct contact with skin and eyes.
- Dilute bases cautiously: Always add the base to water, never water to the base, to prevent splashing and potential burns.
- Dispose of bases properly: Follow local regulations for the safe disposal of chemical waste.
Conclusion
Bases are fundamental chemical species with diverse properties and applications. Understanding their behavior, from their interactions with acids to their industrial uses, is crucial in numerous scientific and technological fields. This comprehensive exploration provides a solid foundation for appreciating the multifaceted nature of bases and their importance in chemistry and beyond. Remember to always prioritize safety when working with bases due to their potential corrosive nature. Further investigation into specific bases and their applications can lead to a deeper and more specialized understanding of this essential area of chemistry.
Latest Posts
Latest Posts
-
How Many Inches Is 20 5 Cm
Apr 06, 2025
-
Cuanto Es 23 Centimetros En Pulgadas
Apr 06, 2025
-
Which Of The Following Must Occur For Speciation To Happen
Apr 06, 2025
-
Which Bacteria Cause The Greatest Harm To The Industry
Apr 06, 2025
-
Cuanto Son 100 Millas En Kilometros
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Is A Property Of A Base . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
