What Is The Electron Configuration For Te
Kalali
Apr 01, 2025 · 6 min read
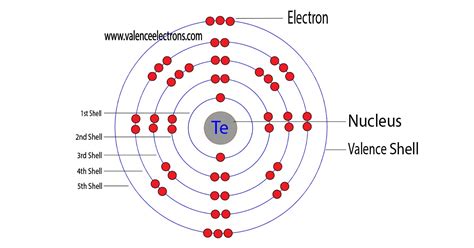
Table of Contents
- What Is The Electron Configuration For Te
- Table of Contents
- What is the Electron Configuration for Te? Understanding Tellurium's Atomic Structure
- Defining Electron Configuration
- Determining Tellurium's Electron Configuration
- Noble Gas Configuration for Tellurium
- Valence Electrons and Chemical Behavior
- Tellurium's Properties and Applications - A Consequence of its Electron Configuration
- Semiconductor Properties:
- Thermoelectric Properties:
- Use in Alloys:
- Catalytic Applications:
- Photovoltaic Applications:
- Rubber Vulcanization:
- Comparing Tellurium to Other Elements in Group 16
- Advanced Concepts and Further Exploration
- Latest Posts
- Latest Posts
- Related Post
What is the Electron Configuration for Te? Understanding Tellurium's Atomic Structure
Tellurium (Te), a metalloid element residing in Group 16 of the periodic table, boasts a fascinating atomic structure reflected in its unique electron configuration. Understanding this configuration is crucial to grasping Tellurium's chemical properties, its behavior in various compounds, and its overall role in diverse scientific applications. This comprehensive guide will delve deep into the electron configuration of Tellurium, exploring its implications and providing a solid foundation for further study.
Defining Electron Configuration
Before diving into Tellurium's specifics, let's establish a clear understanding of electron configuration. An electron configuration is the arrangement of electrons in the various energy levels and sublevels within an atom. It dictates how an atom will interact with other atoms, forming chemical bonds and influencing its overall reactivity. This arrangement follows specific rules dictated by quantum mechanics, including the Aufbau principle (filling orbitals from lowest to highest energy), Hund's rule (maximizing unpaired electrons in a subshell), and the Pauli exclusion principle (no two electrons can have the same four quantum numbers).
Determining Tellurium's Electron Configuration
Tellurium's atomic number is 52, meaning it possesses 52 protons and, in its neutral state, 52 electrons. To determine its electron configuration, we systematically fill the electron shells and subshells according to the rules mentioned above. The order of filling is as follows:
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p...
Applying this order, Tellurium's electron configuration is: 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁶5s²4d¹⁰5p⁴.
Let's break this down:
- 1s²: The first shell (n=1) contains one subshell (s), holding a maximum of 2 electrons.
- 2s²2p⁶: The second shell (n=2) contains an s subshell (2 electrons) and a p subshell (6 electrons).
- 3s²3p⁶: The third shell (n=3) similarly contains an s subshell (2 electrons) and a p subshell (6 electrons).
- 4s²3d¹⁰4p⁶: The fourth shell (n=4) presents a more complex arrangement with an s subshell (2 electrons), a d subshell (10 electrons), and a p subshell (6 electrons). Note that the 3d subshell fills after the 4s subshell due to energy level considerations.
- 5s²4d¹⁰5p⁴: The fifth shell (n=5) contains an s subshell (2 electrons), a d subshell (10 electrons), and a p subshell with 4 electrons. This incomplete p subshell is key to Tellurium's chemical properties.
Noble Gas Configuration for Tellurium
A more concise way to represent Tellurium's electron configuration utilizes the noble gas notation. We can replace the inner electron shells with the symbol of the preceding noble gas, Krypton (Kr), which has an electron configuration of 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁶. Therefore, Tellurium's noble gas configuration is: [Kr]5s²4d¹⁰5p⁴. This notation simplifies the representation while maintaining all essential information.
Valence Electrons and Chemical Behavior
The valence electrons are the electrons in the outermost shell, which are most involved in chemical bonding. In Tellurium's case, the valence electrons are those in the 5s and 5p subshells – a total of six electrons (5s²5p⁴). This configuration explains Tellurium's ability to form various compounds, exhibiting oxidation states ranging from -2 to +6. The four electrons in the 5p subshell are particularly active, contributing to Tellurium's diverse bonding patterns.
The incomplete 5p subshell indicates a tendency to gain or share electrons to achieve a stable octet configuration, like the noble gases. This explains Tellurium's ability to form covalent bonds with other nonmetals and ionic bonds with metals.
Tellurium's Properties and Applications - A Consequence of its Electron Configuration
Tellurium's electron configuration is directly responsible for its unique properties and applications:
Semiconductor Properties:
The partially filled 5p subshell contributes to Tellurium's semiconducting behavior. The relatively small energy gap between the valence and conduction bands allows electrons to move more freely under the influence of an electric field, making it suitable for various electronic applications.
Thermoelectric Properties:
Tellurium's ability to convert heat energy into electrical energy and vice versa stems from its electronic structure. This property finds use in thermoelectric generators and coolers.
Use in Alloys:
Adding Tellurium to other metals improves their machinability and other physical properties. The interaction of Tellurium's valence electrons with the metal's electron structure results in modified mechanical strength and corrosion resistance.
Catalytic Applications:
Tellurium and its compounds can act as catalysts in various chemical reactions. The availability of valence electrons allows for interaction with reactants, influencing reaction pathways and rates.
Photovoltaic Applications:
Tellurium-based compounds, especially cadmium telluride (CdTe), are used in thin-film solar cells. The electronic structure of Tellurium enables efficient absorption of sunlight and generation of electricity.
Rubber Vulcanization:
Tellurium is employed in the vulcanization of rubber, a process that improves its elasticity and durability. The interaction between Tellurium's electrons and the polymer chains influences the cross-linking process.
Comparing Tellurium to Other Elements in Group 16
Tellurium's electron configuration allows for a comparative analysis with other elements in Group 16 (chalcogens): oxygen (O), sulfur (S), selenium (Se), and polonium (Po). All share the same valence electron arrangement (ns²np⁴), leading to similar chemical behaviors in certain respects. However, differences arise due to increasing atomic size and shielding effects down the group. Tellurium, being heavier, exhibits metallic character more pronounced than its lighter counterparts.
Advanced Concepts and Further Exploration
- Excited States: While the ground state electron configuration detailed above is the most stable, Tellurium atoms can absorb energy and transition to higher energy levels (excited states). These excited states have different electron configurations and are important in spectroscopic studies.
- Ionization Energies: The energy required to remove an electron from a Tellurium atom is linked to its electron configuration. The outermost electrons have lower ionization energies compared to those in inner shells.
- Electron Affinity: Tellurium's tendency to gain electrons is reflected in its electron affinity, related to the stability gained upon adding an electron to its outer shell.
- Molecular Orbital Theory: A more sophisticated approach to understanding chemical bonding involves molecular orbital theory. This framework explains the interactions between atomic orbitals to form molecular orbitals in Tellurium-containing molecules and compounds.
This detailed exploration of Tellurium's electron configuration provides a solid foundation for comprehending its unique properties and its crucial role in various applications. Understanding the arrangement of electrons within an atom is fundamental to grasping the behavior of elements and their interactions in the fascinating world of chemistry. Further exploration of the concepts mentioned above will enrich your understanding of Tellurium and its position within the periodic table.
Latest Posts
Related Post
Thank you for visiting our website which covers about What Is The Electron Configuration For Te . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
