What Is The Electron Configuration For Tellurium
Kalali
Apr 02, 2025 · 6 min read
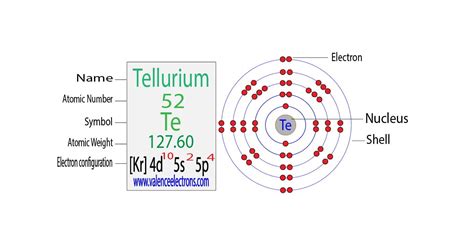
Table of Contents
What is the Electron Configuration for Tellurium? A Deep Dive into Atomic Structure
Tellurium (Te), a metalloid element residing in Group 16 of the periodic table, boasts a fascinating electron configuration that dictates its chemical properties and behavior. Understanding this configuration is key to comprehending its role in various applications, from solar cells to semiconductors. This article delves into the intricacies of Tellurium's electron configuration, exploring its Aufbau principle-based derivation, exceptions, and implications for its reactivity and applications.
Understanding Electron Configuration
Before diving into Tellurium's specific configuration, let's establish a foundational understanding of what electron configuration entails. An electron configuration describes the arrangement of electrons in the various energy levels and sublevels within an atom. This arrangement is governed by several fundamental principles:
The Aufbau Principle
The Aufbau principle, meaning "building-up" in German, dictates that electrons fill atomic orbitals in order of increasing energy levels. Electrons initially occupy the lowest energy levels available, gradually filling higher energy levels as more electrons are added.
Hund's Rule
Hund's rule states that electrons will individually occupy each orbital within a subshell before doubling up in any one orbital. This minimizes electron-electron repulsion and results in a more stable configuration.
Pauli Exclusion Principle
The Pauli exclusion principle asserts that no two electrons within an atom can have the same set of four quantum numbers (n, l, ml, and ms). This means each orbital can accommodate a maximum of two electrons with opposite spins.
Deriving Tellurium's Electron Configuration
Tellurium (Te) has an atomic number of 52, meaning it possesses 52 electrons. Applying the Aufbau principle and Hund's rule, we can systematically determine its electron configuration:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁴
Let's break this down:
- 1s²: Two electrons occupy the first energy level (n=1), specifically the s subshell.
- 2s² 2p⁶: Eight electrons fill the second energy level (n=2), with two in the 2s subshell and six in the 2p subshell.
- 3s² 3p⁶: Eight electrons fill the third energy level (n=3), similarly distributed between the 3s and 3p subshells.
- 4s² 3d¹⁰ 4p⁶: Eighteen electrons populate the fourth energy level (n=4), including two in the 4s, ten in the 3d, and six in the 4p subshells. Note the filling order: 4s fills before 3d.
- 5s² 4d¹⁰ 5p⁴: Sixteen electrons occupy the fifth energy level (n=5), with two in the 5s, ten in the 4d, and four in the 5p subshells. Again, observe the filling order: 5s fills before 4d.
This complete electron configuration showcases the systematic filling of orbitals according to the Aufbau principle. The outermost shell, or valence shell, is the fifth energy level, containing six electrons in the 5s and 5p subshells. These valence electrons are crucial in determining Tellurium's chemical reactivity.
Tellurium's Valence Electrons and Chemical Properties
The four electrons in the 5p subshell are Tellurium's valence electrons. These electrons participate in chemical bonding, influencing its reactivity and ability to form compounds. Having six valence electrons, Tellurium exhibits characteristics typical of Group 16 elements (chalcogens):
- Covalent bonding: Tellurium readily forms covalent bonds with other nonmetals by sharing its valence electrons. This is evident in compounds like tellurium dioxide (TeO₂) and hydrogen telluride (H₂Te).
- Variable oxidation states: Due to its relatively accessible p-orbital electrons, Tellurium can exhibit various oxidation states, including -2, +2, +4, and +6. This versatility allows it to participate in a wide range of chemical reactions.
- Amphoteric nature: Tellurium displays amphoteric behavior, meaning it can react with both acids and bases. This arises from its ability to act as both an electron donor and an electron acceptor.
Orbital Diagrams and Electron Spin
A more detailed representation of Tellurium's electron configuration utilizes orbital diagrams. These diagrams illustrate the individual orbitals within each subshell and the electron occupancy within them, including electron spin. For example, the 5p subshell (5p⁴) would be depicted with three orbitals, each capable of holding two electrons with opposite spins (↑↓). In Tellurium's case, the four electrons would occupy these orbitals according to Hund's rule, with two orbitals containing one electron each and the third orbital containing a pair of electrons.
Exceptions to the Aufbau Principle: The Case of Tellurium
While the Aufbau principle provides a general guideline for electron configuration, certain exceptions exist. Tellurium's configuration, while following the general rule, hints at subtle deviations. These subtle deviations are better explained using more advanced quantum mechanical calculations that go beyond the simple Aufbau principle.
The seemingly straightforward application of the Aufbau principle leads to the expected configuration. However, more precise calculations and experimental observations reveal that the energies of the 5s and 4d orbitals are closer than predicted by a simple Aufbau approach. This proximity can influence electron placement and potentially lead to slight variations. These nuances are often reflected in spectroscopic data and chemical behavior.
Applications of Tellurium and its Electron Configuration
Tellurium's unique electron configuration translates into several valuable applications:
- Semiconductors: Tellurium's semiconducting properties stem directly from its electron configuration and the availability of valence electrons for electrical conduction under specific conditions. This makes it useful in various electronic devices and solar cells.
- Alloying agent: Tellurium's ability to form alloys with metals improves their machinability and enhances their resistance to corrosion.
- Inorganic pigments: Tellurium compounds are used as pigments in paints and plastics due to their distinctive colors.
- Catalysis: Tellurium compounds find limited use as catalysts in certain chemical reactions.
- Rubber vulcanization: Tellurium is sometimes employed in the vulcanization process of rubber, though less commonly than sulfur.
Conclusion: Tellurium's Electron Configuration in Context
The electron configuration of Tellurium (1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁴) is not just an abstract arrangement of electrons; it is the fundamental blueprint that dictates its chemical and physical properties. Understanding this configuration is paramount for appreciating Tellurium's role in various applications and for predicting its reactivity. While the Aufbau principle serves as a valuable tool for predicting electron configurations, subtle nuances and exceptions, as hinted at in Tellurium's case, remind us that more sophisticated quantum mechanical treatments are needed for a fully comprehensive understanding of electronic structure and properties. Further research and advanced theoretical studies continue to refine our understanding of these complex interactions, providing a richer and more complete picture of Tellurium's fascinating atomic structure and its consequential applications. The seemingly simple electron configuration is thus revealed as a window into a complex world of atomic interactions, highlighting the intricate interplay of fundamental principles and observable phenomena.
Latest Posts
Latest Posts
-
12 Out Of 20 As Percentage
Apr 03, 2025
-
How Many Meters In 2 Km
Apr 03, 2025
-
What Percentage Of 50 Is 30
Apr 03, 2025
-
How Many Liters Is 8 Gallons
Apr 03, 2025
-
64 Oz Is How Many Cups
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Electron Configuration For Tellurium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
