What Is The General Chemical Equation For An Endothermic Reaction
Kalali
Apr 03, 2025 · 5 min read
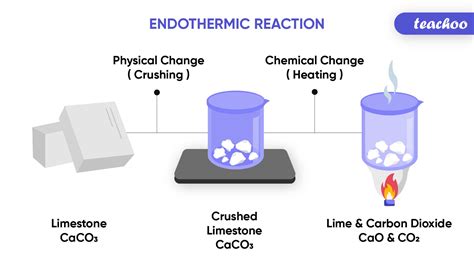
Table of Contents
What is the General Chemical Equation for an Endothermic Reaction?
Endothermic reactions are a fascinating facet of chemistry, representing processes where energy is absorbed from the surroundings. Understanding these reactions requires delving into their fundamental nature, represented by their chemical equations and the thermodynamic principles governing them. This comprehensive article will explore the concept of endothermic reactions, dissect their general chemical equations, delve into examples, and discuss their significance in various fields.
Defining Endothermic Reactions: An Energy Perspective
Before we dive into the chemical equations, let's solidify our understanding of what constitutes an endothermic reaction. In essence, an endothermic reaction is a chemical or physical process that absorbs heat energy from its surroundings. This absorption of energy leads to a decrease in the temperature of the immediate environment. Imagine a cold pack used for injuries – the pack cools down because the chemical reaction inside is endothermic, drawing heat away from its surroundings.
This energy absorption is reflected in the enthalpy change (ΔH) of the reaction. Enthalpy is a thermodynamic property representing the total heat content of a system. In an endothermic reaction, the ΔH is positive, indicating that the products possess higher enthalpy than the reactants. This difference in enthalpy represents the energy absorbed during the reaction.
The General Chemical Equation: Representing Energy Absorption
Unlike a simple stoichiometric equation that merely shows the reactant and product ratios, representing an endothermic reaction requires explicitly showing the energy absorption. While there isn't a single, universally accepted "general" chemical equation format that incorporates energy, we can represent it symbolically. The general approach involves including + heat or + energy on the reactant side of the equation.
For a generic endothermic reaction involving reactants A and B forming products C and D, the general chemical equation can be written as:
A + B + heat → C + D
Or, equivalently:
A + B + energy → C + D
This notation clearly indicates that heat or energy is required as a reactant for the reaction to proceed. This contrasts with exothermic reactions, where heat is released as a product.
Important Note: The actual amount of heat absorbed (in Joules or Kilojoules) can be quantitatively represented by including the specific ΔH value within the equation. However, the general equation focuses on the qualitative aspect of energy absorption rather than the quantitative measurement.
Examples of Endothermic Reactions: From Everyday Life to Industrial Processes
Let's illustrate this concept with some concrete examples of endothermic reactions, showcasing their diversity across various applications:
1. Photosynthesis: The Foundation of Life
One of the most crucial endothermic processes on Earth is photosynthesis. Plants absorb sunlight (energy) to convert carbon dioxide and water into glucose (a sugar) and oxygen. The general equation is:
6CO₂ + 6H₂O + sunlight → C₆H₁₂O₆ + 6O₂
Here, sunlight acts as the energy input, making it a classic example of an endothermic reaction.
2. Melting Ice: A Phase Transition
The melting of ice is a physical change, not a chemical reaction, yet it perfectly exemplifies an endothermic process. Energy (heat) is required to break the hydrogen bonds holding the water molecules in the solid (ice) state, transforming them into the liquid state (water). The equation isn't strictly a chemical equation, but can be represented as:
H₂O(s) + heat → H₂O(l)
3. Dissolving Ammonium Nitrate in Water: A Common Demonstration
Dissolving ammonium nitrate (NH₄NO₃) in water is a readily demonstrable endothermic reaction. The process absorbs heat from the surrounding water, causing a noticeable temperature drop. The equation is:
NH₄NO₃(s) + H₂O(l) + heat → NH₄⁺(aq) + NO₃⁻(aq)
This reaction is commonly used in instant cold packs for injuries.
4. Decomposition of Calcium Carbonate: Industrial Applications
The decomposition of calcium carbonate (limestone) into calcium oxide (lime) and carbon dioxide requires a significant input of heat, making it an industrial example of an endothermic process:
CaCO₃(s) + heat → CaO(s) + CO₂(g)
This reaction is crucial in the cement industry, where high temperatures are used to drive the decomposition.
5. Cooking an Egg: A Complex Endothermic Process
While the cooking of an egg seems simple, it involves multiple endothermic and exothermic reactions. The denaturation of proteins, a key part of cooking, absorbs energy as the protein molecules unfold. The full complexity of the process makes a simple chemical equation impractical, but the overall effect is heat absorption, making it an endothermic process.
Factors Influencing Endothermic Reactions: Reaction Rates and Equilibrium
Several factors can affect the rate and equilibrium of endothermic reactions:
-
Temperature: Increasing the temperature generally increases the rate of an endothermic reaction, as more energy is available to overcome the activation energy barrier. However, the effect on equilibrium is less predictable and depends on the specific reaction.
-
Concentration: Increasing the concentration of reactants generally increases the rate of reaction but has a less defined effect on equilibrium.
-
Surface Area: For reactions involving solids, increasing the surface area of the reactants can increase the reaction rate. This is because more reactant molecules are exposed to the environment.
-
Catalysts: Catalysts can increase the rate of an endothermic reaction by lowering the activation energy, facilitating the reaction without being consumed themselves. However, they do not affect the equilibrium position.
The Importance of Endothermic Reactions in Various Fields
Endothermic reactions play crucial roles in a variety of fields:
-
Industry: Many industrial processes, such as the production of metals from their ores and the manufacture of certain chemicals, rely on endothermic reactions.
-
Biology: Photosynthesis is the ultimate example of endothermic reactions sustaining life on Earth. Endothermic processes are also vital in numerous metabolic reactions within organisms.
-
Medicine: Endothermic reactions are exploited in cold packs for injury treatment and certain types of drug delivery systems.
-
Environmental Science: Understanding endothermic reactions is crucial for studying climate change and other environmental processes.
Conclusion: Endothermic Reactions – A Fundamental Aspect of Chemistry
Endothermic reactions represent a fundamental class of chemical and physical processes characterized by their absorption of energy from their surroundings. While a single, universally applicable general chemical equation doesn't exist, representing the energy input as "+ heat" or "+ energy" on the reactant side effectively communicates this essential feature. The examples provided demonstrate the broad impact of endothermic processes in various fields, underscoring their importance in both natural and technological contexts. Understanding these reactions is key to advancements in various disciplines, from sustainable energy solutions to medical therapies.
Latest Posts
Latest Posts
-
166 Cm To Feet And Inches
Apr 04, 2025
-
1 1 2 Cup Of Water
Apr 04, 2025
-
How Much Is 10 Fl Oz
Apr 04, 2025
-
12 Ounces Of Water Is How Many Cups
Apr 04, 2025
-
Is Food Coloring In Water A Chemical Change
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The General Chemical Equation For An Endothermic Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
