Why Do All Enzymatic Reactions Need Activation Energy
Kalali
Apr 05, 2025 · 6 min read
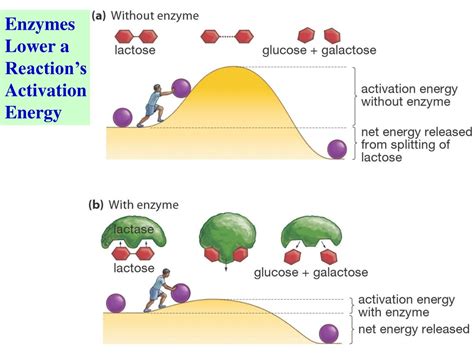
Table of Contents
Why Do All Enzymatic Reactions Need Activation Energy?
Enzymes are biological catalysts that significantly speed up the rate of virtually all chemical reactions within cells. Despite their accelerating effect, enzymes don't actually change the overall thermodynamics of a reaction—meaning they don't alter the free energy difference between reactants and products. Instead, they lower the activation energy (Ea), the energy barrier that must be overcome for a reaction to proceed. This crucial concept is fundamental to understanding how enzymes function and why they are essential for life. This article delves into the reasons why activation energy is necessary for even enzyme-catalyzed reactions, exploring the underlying principles of chemical kinetics and the multifaceted roles of enzymes.
The Nature of Activation Energy
Before examining the role of activation energy in enzymatic reactions, it's crucial to understand what activation energy is. Chemical reactions, at their core, involve the breaking and forming of chemical bonds. Even seemingly spontaneous reactions require an initial input of energy to initiate the bond-breaking process. This initial energy input is the activation energy.
Think of it like pushing a boulder over a hill. The boulder represents the reactants, the hill represents the activation energy barrier, and the valley on the other side represents the products. You need to put in energy (push the boulder) to get it over the hill, even if the valley is lower in energy than the starting point. Once the boulder is over the hill, it rolls down spontaneously. Similarly, once reactants overcome the activation energy barrier, the reaction proceeds spontaneously towards the formation of products.
The Transition State
The activation energy is closely related to the transition state. The transition state is a high-energy, unstable intermediate state that exists briefly during the conversion of reactants to products. It's the point of maximum energy along the reaction coordinate. The activation energy is the difference in energy between the reactants and the transition state.
Reaching the transition state is the rate-limiting step of the reaction. The higher the activation energy, the slower the reaction rate because fewer molecules will possess the necessary energy to reach the transition state. This is why reactions with high activation energies tend to be slow at ambient temperatures.
Enzymes and the Reduction of Activation Energy
Enzymes dramatically accelerate reaction rates by lowering the activation energy. They achieve this by a variety of mechanisms, all centered around facilitating the formation of the transition state. Despite this reduction, activation energy remains a necessity, even in the presence of enzymes. This necessity stems from several key factors:
1. The Nature of Chemical Bonds:
The fundamental reason why activation energy is needed, even with enzymes, lies in the nature of chemical bonds. Breaking existing bonds and forming new ones requires energy. Enzymes, while highly efficient, cannot circumvent the fundamental laws of chemistry governing bond breaking and formation. They merely provide a more efficient pathway, lowering the energy required to reach the transition state.
Enzymes do this by precisely orienting the reactants, inducing strain in existing bonds, or providing alternative reaction pathways with lower activation energies, but bond manipulation itself requires energy input.
2. The Transition State's Instability:
The transition state itself is inherently unstable. It's a high-energy, fleeting intermediate, existing only for a very short time. The very nature of its instability demands energy input to form it. Enzymes stabilize the transition state, making it slightly less unstable and thus reducing the energy required to reach it. However, reaching even this stabilized transition state still requires an input of energy.
3. The Importance of Molecular Collisions with Sufficient Energy:
Even with reduced activation energy, the reaction still relies on successful molecular collisions with sufficient energy. Enzymes increase the likelihood of successful collisions by orienting reactants correctly and by providing a favorable microenvironment. However, the molecules still need to possess a certain minimum amount of kinetic energy to overcome the remaining activation energy barrier, no matter how low it is. Without this energy, the reaction will simply not proceed, even with the enzyme's assistance.
4. Maintaining Thermodynamic Equilibrium:
Enzymes do not alter the overall thermodynamics of a reaction; they do not affect the difference between the free energies of the reactants and products. Therefore, if the activation energy was completely eliminated, it would violate the principles of thermodynamics and cause spontaneous reactions to proceed at infinitely fast rates. This would disrupt cellular homeostasis and equilibrium. By maintaining a reduced, yet non-zero, activation energy, enzymes maintain a controlled reaction rate that's essential for cellular function.
Specific Mechanisms by Which Enzymes Lower Activation Energy
Enzymes employ various ingenious strategies to lower the activation energy. These include:
-
Substrate Binding and Orientation: Enzymes bind their substrates with high specificity, positioning them precisely to facilitate bond breaking and formation. This precise orientation reduces the need for random collisions with the appropriate energy and orientation.
-
Induced Fit: The binding of a substrate often induces conformational changes in the enzyme, further optimizing the interaction and lowering the activation energy. This dynamic interaction is crucial for many enzyme-catalyzed reactions.
-
Acid-Base Catalysis: Enzyme active sites often contain amino acid residues that can act as acids or bases, donating or accepting protons to facilitate bond breakage or formation.
-
Covalent Catalysis: In some cases, the enzyme forms a temporary covalent bond with the substrate, forming a reaction intermediate that is more readily converted to the product.
-
Metal Ion Catalysis: Many enzymes use metal ions to stabilize negative charges, facilitate redox reactions, or bind substrates.
Consequences of a Lack of Activation Energy
If enzymatic reactions did not require activation energy, the consequences would be catastrophic for life. Imagine a situation where all metabolic processes proceeded at infinitely fast rates. This uncontrolled reactivity would lead to:
-
Cellular Chaos: The highly regulated and precisely timed reactions within a cell would become completely disorganized. Metabolic pathways would not be able to function efficiently.
-
Energy Waste: Energy would be expended without control, leading to excessive heat production and potentially cell damage.
-
Loss of Regulation: Cells would lose the ability to control the rate of their reactions, making it impossible to respond to environmental changes or maintain homeostasis.
-
Unwanted Side Reactions: The increased reaction rate would make unwanted side reactions far more likely, producing harmful byproducts and disrupting cellular functions.
In essence, the absence of activation energy would render life, as we know it, impossible.
Conclusion: The Indispensable Role of Activation Energy in Enzymatic Reactions
In summary, while enzymes dramatically accelerate reaction rates by reducing activation energy, they cannot entirely eliminate it. The need for activation energy is intrinsic to the fundamental nature of chemical bonds, the instability of the transition state, and the principles of thermodynamics that govern all chemical processes. Activation energy ensures controlled and regulated reaction rates, preventing cellular chaos and maintaining the delicate balance necessary for life. Enzymes, through their diverse catalytic mechanisms, effectively lower this energy barrier, allowing life’s complex biochemical processes to proceed efficiently and maintain the integrity and functionality of living organisms. The careful balance between facilitating reactions and maintaining the requirement for activation energy underscores the exquisite precision and efficiency of enzyme catalysis.
Latest Posts
Latest Posts
-
What Percent Is 17 Of 25
Apr 06, 2025
-
25 Centimeters Equals How Many Inches
Apr 06, 2025
-
How Many Mm In 20 Cm
Apr 06, 2025
-
Find 4 Consecutive Integers With The Sum Of 54
Apr 06, 2025
-
50 Mg Is How Many Grams
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Why Do All Enzymatic Reactions Need Activation Energy . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
