An Atom That Has Gained An Electron Is Called
Kalali
Apr 05, 2025 · 6 min read
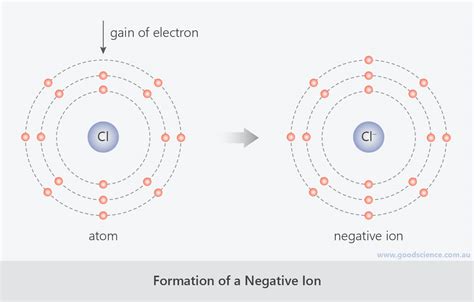
Table of Contents
An Atom That Has Gained an Electron is Called an Anion: A Deep Dive into Ionic Bonds and Chemical Reactions
Have you ever wondered what happens when an atom, the fundamental building block of matter, acquires an extra electron? The answer is simple yet profound: it becomes an anion. This seemingly small event has significant consequences, influencing the atom's properties, its interactions with other atoms, and ultimately shaping the world around us. This article delves into the fascinating world of anions, exploring their formation, properties, and crucial role in chemical reactions and the larger framework of chemistry.
Understanding Ions: The Foundation of Anion Formation
Before we dive into the specifics of anions, let's establish a fundamental understanding of ions. An ion is an atom or molecule that carries a net electrical charge. This charge arises from an imbalance between the number of protons (positively charged particles) and electrons (negatively charged particles) within the atom or molecule. There are two primary types of ions:
Cations: Positively Charged Ions
Cations are formed when an atom loses one or more electrons. This loss leaves the atom with more protons than electrons, resulting in a net positive charge. For example, a sodium atom (Na) readily loses one electron to become a sodium ion (Na⁺).
Anions: Negatively Charged Ions
Conversely, anions are formed when an atom gains one or more electrons. This gain results in an atom possessing more electrons than protons, leading to a net negative charge. A classic example is a chlorine atom (Cl) gaining one electron to become a chloride ion (Cl⁻).
The Process of Anion Formation: Electron Affinity and Electronegativity
The ability of an atom to gain an electron and form an anion is determined by two key properties: electron affinity and electronegativity.
Electron Affinity: The Atom's Attraction to Electrons
Electron affinity refers to the energy change that occurs when an atom gains an electron. A high electron affinity indicates that the atom strongly attracts electrons, making it more likely to form an anion. Halogens, for instance, possess high electron affinities, readily accepting an electron to achieve a stable electron configuration.
Electronegativity: The Atom's Pull on Shared Electrons
Electronegativity measures an atom's ability to attract electrons within a chemical bond. While electron affinity focuses on an isolated atom gaining an electron, electronegativity considers the attraction within a bond. Atoms with high electronegativity tend to attract electrons strongly, often resulting in the formation of anions when bonding with less electronegative atoms.
The Octet Rule and Anion Stability
The stability of an anion is often explained by the octet rule. This rule states that atoms tend to gain, lose, or share electrons to achieve a full outer electron shell containing eight electrons (except for hydrogen and helium, which aim for two). By gaining electrons, atoms can complete their outer shell, achieving a more stable electron configuration similar to that of noble gases, which are notoriously unreactive.
For example, oxygen (O) has six electrons in its outer shell. By gaining two electrons, it becomes an oxide ion (O²⁻), completing its octet and achieving a stable, noble gas-like electron configuration.
Examples of Common Anions
Many common ions found in everyday life and chemical reactions are anions. Here are a few examples:
- Chloride ion (Cl⁻): Found in table salt (NaCl) and many other compounds.
- Sulfide ion (S²⁻): Found in minerals like pyrite (iron sulfide).
- Oxide ion (O²⁻): A crucial component of many metal oxides and minerals.
- Nitrate ion (NO₃⁻): A vital nutrient in fertilizers and a common component in many chemical processes.
- Phosphate ion (PO₄³⁻): Essential for biological processes, including energy transfer (ATP) and DNA structure.
- Hydroxide ion (OH⁻): Involved in acid-base reactions and found in alkaline solutions.
- Carbonate ion (CO₃²⁻): Found in limestone and plays a crucial role in the carbon cycle.
The Role of Anions in Ionic Bonding
Anions are key players in ionic bonding, a type of chemical bond formed through the electrostatic attraction between oppositely charged ions. When an atom with a low electronegativity (like a metal) loses electrons to form a cation, and an atom with a high electronegativity (like a non-metal) gains electrons to form an anion, the resulting opposite charges attract each other, creating a strong ionic bond. This interaction forms ionic compounds like sodium chloride (NaCl), where the sodium cation (Na⁺) and chloride anion (Cl⁻) are held together by electrostatic forces.
The Importance of Anions in Chemical Reactions
Anions are not merely passive participants; they actively contribute to a wide array of chemical reactions. Their presence significantly affects the reaction rate, product formation, and overall reaction pathway.
Acid-Base Reactions: Anions as Conjugate Bases
In acid-base reactions, anions often act as conjugate bases. When an acid donates a proton (H⁺), the remaining part of the acid becomes an anion, which can accept a proton back, acting as a base. For example, when hydrochloric acid (HCl) donates a proton, it forms a chloride anion (Cl⁻), which can act as a conjugate base.
Precipitation Reactions: Formation of Insoluble Salts
Anions also play a key role in precipitation reactions. When two solutions containing soluble salts are mixed, an insoluble ionic compound (precipitate) can form. This often involves the combination of specific cations and anions to create a compound that is insoluble in the solvent. For example, the reaction between silver nitrate (AgNO₃) and sodium chloride (NaCl) leads to the formation of the insoluble silver chloride (AgCl) precipitate.
Redox Reactions: Anions as Electron Acceptors
Anions can participate in redox (reduction-oxidation) reactions. Anions often act as electron acceptors (oxidants), gaining electrons during the reaction. For example, the reduction of oxygen (O₂) to form oxide ions (O²⁻) involves the oxygen molecule gaining electrons.
Anions in Biological Systems
Anions are crucial for life. They play essential roles in various biological processes:
- Phosphate ions (PO₄³⁻): Fundamental components of DNA and RNA, essential for genetic information storage and transfer. Also vital for energy transfer in ATP (adenosine triphosphate).
- Bicarbonate ions (HCO₃⁻): Important for maintaining blood pH balance.
- Chloride ions (Cl⁻): Involved in nerve impulse transmission.
- Various organic anions: Crucial components of many biological molecules, such as amino acids and fatty acids.
Analyzing Anions: Techniques and Applications
Identifying and analyzing anions is vital in many scientific fields. Several techniques are used:
- Spectroscopy: Techniques like infrared (IR) spectroscopy and nuclear magnetic resonance (NMR) spectroscopy provide valuable information about the structure and composition of anions.
- Chromatography: Different types of chromatography, such as ion chromatography, are used to separate and analyze mixtures of anions.
- Electrochemical methods: Techniques like potentiometry and voltammetry are used to determine the concentration of anions in solutions.
Conclusion: The Unsung Heroes of Chemistry
Anions, often overlooked, are fundamental to our understanding of chemistry and the world around us. From forming ionic compounds to participating in crucial biological processes, their influence is undeniable. Their role in chemical reactions, their presence in biological systems, and the sophisticated techniques used to analyze them highlight the critical importance of these negatively charged ions in shaping the chemical landscape of our universe. Understanding their properties and behavior allows scientists to design and develop new materials, understand complex biological processes, and contribute to advancements across various scientific disciplines. The seemingly simple act of an atom gaining an electron leads to a cascade of consequences, demonstrating the power and elegance of fundamental chemical principles.
Latest Posts
Latest Posts
-
Contains More Oh Ions Than H Ions
Apr 05, 2025
-
What Is The Least Common Multiple Of 10 And 2
Apr 05, 2025
-
How Many Inches Is 101 Cm
Apr 05, 2025
-
How Many Ounces Is A Third Of A Pound
Apr 05, 2025
-
What Is The Lcm Of 8 And 9
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about An Atom That Has Gained An Electron Is Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
