Baking Soda An Acid Or Base
Kalali
Apr 05, 2025 · 5 min read
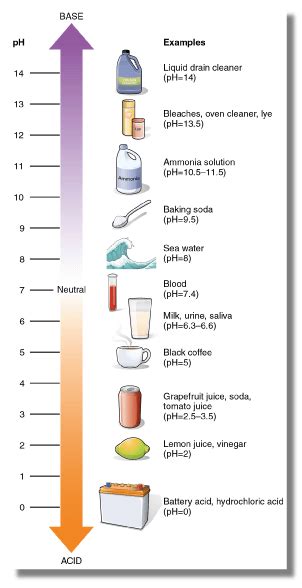
Table of Contents
Baking Soda: An Acid or a Base? Understanding its Chemical Nature
Baking soda, a common household ingredient, is often the subject of confusion when it comes to its chemical nature. Is it an acid or a base? The answer, while seemingly simple, requires a deeper dive into chemistry to fully understand. This comprehensive guide will explore the chemical properties of baking soda, its reactions with acids and bases, and its various applications, ultimately clarifying its role in the world of chemistry and cooking.
What is Baking Soda?
Baking soda, also known as sodium bicarbonate (NaHCO₃), is a white crystalline powder. It's a chemical compound that's naturally occurring but also widely manufactured. Its unique properties stem from its amphoteric nature—a characteristic that allows it to act as both an acid and a base depending on the environment.
The Chemistry of Baking Soda: A Closer Look
To grasp baking soda's behavior, we need to understand the concept of pH. The pH scale measures how acidic or basic a substance is, ranging from 0 to 14. A pH of 7 is neutral; below 7 is acidic, and above 7 is basic (or alkaline). Baking soda's pH in water is around 8.3, placing it firmly in the basic range. This is due to its ability to accept protons (H⁺ ions) from an acid.
The bicarbonate ion (HCO₃⁻) in baking soda is the key player in its reactivity. This ion can act as both a proton acceptor (base) and a proton donor (acid), showcasing its amphoteric nature.
Baking Soda as a Base: Reactions with Acids
Baking soda's most well-known characteristic is its reaction with acids. This reaction is the basis of its use in baking and other applications. When baking soda reacts with an acid, it undergoes a neutralization reaction, producing carbon dioxide (CO₂), water (H₂O), and a salt. This CO₂ gas is what causes the leavening effect in baking, making baked goods rise.
Here’s a simplified representation of the reaction with a common acid, acetic acid (found in vinegar):
NaHCO₃ (baking soda) + CH₃COOH (acetic acid) → CH₃COONa (sodium acetate) + H₂O (water) + CO₂ (carbon dioxide)
This reaction is crucial in baking recipes involving acidic ingredients like buttermilk, lemon juice, or vinegar. The interaction produces the necessary CO₂ bubbles for a light and airy texture. The type of acid used influences the flavor and rate of the reaction.
Examples of Acid-Base Reactions with Baking Soda:
- Baking: The reaction with acids in ingredients like buttermilk or yogurt creates the leavening action in cakes, muffins, and quick breads.
- Antacid: Baking soda can neutralize stomach acid, providing temporary relief from heartburn. However, frequent use is not recommended, and medical advice should be sought for persistent heartburn.
- Cleaning: Its reaction with acidic substances like vinegar can help clean and deodorize surfaces. The fizzing action helps loosen dirt and grime.
Baking Soda's Behavior in Different Environments
The reaction of baking soda isn't always straightforward. The presence of other ingredients and environmental factors can affect its behavior.
The Role of Moisture
Moisture is essential for the acid-base reaction to occur effectively. Dry baking soda and a dry acid won't react significantly. The presence of water facilitates the dissociation of the baking soda and the acid, allowing the ions to interact and produce CO₂.
The Impact of Temperature
Temperature also plays a role. Higher temperatures generally speed up the reaction rate, leading to quicker CO₂ production. This is why baking soda is typically added to a recipe after other ingredients are combined. Premature activation can lead to a flat baked good.
Interaction with Other Ingredients
Other ingredients in a recipe can influence the rate and extent of the reaction. The presence of certain salts or other buffering agents can modify the pH and affect the overall outcome.
Baking Soda vs. Baking Powder: Key Differences
Baking powder is often confused with baking soda, but they are distinct. Baking powder contains baking soda along with an acid (usually cream of tartar) and a drying agent (usually cornstarch). This means baking powder is a complete leavening agent, containing everything needed for a reaction, while baking soda requires an external acid source.
Baking soda needs an acid; baking powder doesn't.
Baking Soda’s Other Applications: Beyond Baking
The versatility of baking soda extends far beyond the kitchen:
- Cleaning: It's a powerful natural cleaner, effective in removing stains, grease, and odors from various surfaces. Its abrasive nature and ability to neutralize acids make it a multi-purpose cleaning agent.
- Deodorizing: Baking soda absorbs odors effectively. It's often used in refrigerators, shoes, and carpets to neutralize unpleasant smells.
- Personal Care: It can be used as a mild abrasive in toothpaste, helping to clean teeth and whiten them naturally. It can also be used in homemade deodorants.
- Gardening: It can help adjust the soil pH, improving its suitability for specific plants.
Potential Drawbacks and Safety Considerations
While baking soda is generally safe, excessive or inappropriate use can have some drawbacks:
- Ingestion: Consuming large amounts of baking soda can disrupt the body's acid-base balance (electrolyte imbalance), leading to potential health problems.
- Skin Irritation: Some individuals may experience skin irritation or allergic reactions from prolonged contact.
- Inhalation: Inhaling baking soda powder can irritate the lungs.
Conclusion: Baking Soda – A Versatile Amphoteric Compound
Baking soda's amphoteric nature allows it to act as both an acid and a base, giving it a wide array of applications. Its reaction with acids produces carbon dioxide gas, a fundamental principle behind its leavening power in baking. Understanding its chemical properties and interactions with other substances is crucial for both safe and effective use in various applications, from cooking and cleaning to personal care and gardening. Its versatility and relatively safe nature make it a truly indispensable household item. However, always use it responsibly, following instructions and exercising caution to avoid potential drawbacks. Remembering its basic nature (pun intended!) will help you understand its behavior and utilize its capabilities fully. Furthermore, always consult with professionals for medical advice or specific technical applications.
Latest Posts
Latest Posts
-
How Many Centimeters Are In 2 Meters
Apr 05, 2025
-
15 Percent Of 80 Is What Number
Apr 05, 2025
-
Common Multiples Of 4 5 And 6
Apr 05, 2025
-
What Is 17 Out Of 25
Apr 05, 2025
-
How Many Grams Are 3 Oz
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Baking Soda An Acid Or Base . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
