Calcium Is A Metal Nonmetal Or Metalloid
Kalali
Apr 05, 2025 · 6 min read
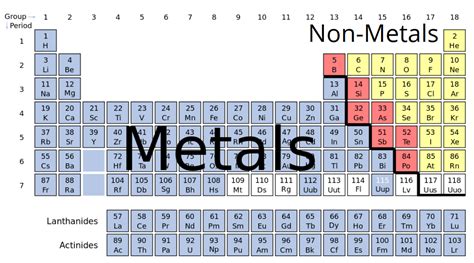
Table of Contents
Calcium: Metal, Nonmetal, or Metalloid? A Deep Dive into its Properties
Calcium is a crucial element for life, playing a vital role in everything from bone structure to nerve function. But what about its fundamental chemical nature? Is calcium a metal, nonmetal, or metalloid? The answer is clear-cut: calcium is a metal. This article will delve into the properties that definitively classify calcium as a metal and explore its various characteristics, applications, and significance in biological systems.
Understanding the Classification of Elements
Before we dive into the specifics of calcium, let's briefly review the classification of elements into metals, nonmetals, and metalloids. This classification is primarily based on an element's properties, specifically its:
- Electrical Conductivity: Metals are excellent conductors of electricity, while nonmetals are generally poor conductors. Metalloids exhibit intermediate conductivity.
- Thermal Conductivity: Similar to electrical conductivity, metals are good conductors of heat, whereas nonmetals are poor conductors. Metalloids again show intermediate behavior.
- Malleability and Ductility: Metals are malleable (can be hammered into sheets) and ductile (can be drawn into wires), while nonmetals are typically brittle.
- Luster: Metals usually possess a shiny, metallic luster, unlike nonmetals, which lack this characteristic.
- Ionization Energy and Electronegativity: Metals tend to have low ionization energies (easily lose electrons) and low electronegativities (weakly attract electrons). Nonmetals have high ionization energies and high electronegativities.
Calcium: A Definitive Metal
Calcium unequivocally fits the profile of a metal. Let's examine its properties in detail:
1. Physical Properties of Calcium: Solid Evidence of Metallic Nature
- Appearance: Calcium is a silvery-white, relatively soft metal. Its metallic luster is readily observable in its pure form, although it tarnishes quickly in air due to oxidation.
- State at Room Temperature: Calcium exists as a solid at room temperature, a common characteristic of metals.
- Electrical and Thermal Conductivity: Calcium is a good conductor of both electricity and heat, further supporting its metallic classification. This high conductivity stems from the free movement of electrons in its metallic structure.
- Malleability and Ductility: Although less malleable and ductile than some other metals like gold or copper, calcium still exhibits these properties to a significant degree. It can be hammered into sheets and drawn into wires, albeit with some difficulty. This malleability and ductility are directly linked to its metallic bonding.
- Density: Calcium has a relatively low density compared to many other metals, but its density is still higher than that of many nonmetals.
2. Chemical Properties of Calcium: The Reactivity of a Metal
Calcium's chemical properties strongly reinforce its classification as a metal.
- Low Ionization Energy: Calcium readily loses its two outermost electrons to achieve a stable electron configuration. This low ionization energy is a hallmark of metals, reflecting their tendency to form positive ions (cations).
- Low Electronegativity: Calcium has a low electronegativity, indicating its weak attraction for electrons. This contrasts sharply with nonmetals, which have high electronegativities and tend to gain electrons to form negative ions (anions).
- Reactivity with Water and Acids: Calcium is highly reactive with water and acids, producing hydrogen gas. This reactivity is typical of many reactive metals. The reaction with water is exothermic, meaning it releases heat.
- Oxidation: Calcium readily oxidizes in air, forming calcium oxide (CaO). This oxidation process is a characteristic chemical reaction for many metals.
- Formation of Ionic Compounds: Calcium readily forms ionic compounds with nonmetals. In these compounds, calcium loses electrons to become a positively charged ion (Ca²⁺), while the nonmetal gains electrons to become a negatively charged ion. The electrostatic attraction between these oppositely charged ions forms the ionic bond.
3. Crystal Structure: The Ordered Arrangement of Metal Atoms
Calcium possesses a face-centered cubic (FCC) crystal structure. This is a common crystal structure for many metals, characterized by a highly ordered arrangement of atoms. This ordered arrangement contributes to the physical properties of calcium, such as its malleability and ductility. The close packing of atoms in the FCC structure allows for the easy slippage of atomic planes past one another, resulting in the ability to deform without breaking.
Calcium's Biological Significance: An Essential Metal for Life
Calcium's importance extends far beyond its chemical classification. It's an essential mineral for all living organisms, playing a crucial role in numerous biological processes:
- Bone and Tooth Formation: Calcium is the major structural component of bones and teeth, providing strength and support to the skeletal system. Hydroxyapatite, a calcium phosphate mineral, forms the crystalline structure of bone.
- Muscle Contraction: Calcium ions (Ca²⁺) are essential for muscle contraction. The release of Ca²⁺ ions triggers the interaction between actin and myosin filaments, leading to muscle shortening.
- Nerve Impulse Transmission: Calcium ions play a critical role in nerve impulse transmission. The influx of Ca²⁺ ions into nerve cells triggers the release of neurotransmitters, allowing for communication between nerve cells.
- Blood Clotting: Calcium is a necessary cofactor in the blood clotting process. It participates in a complex cascade of reactions that ultimately lead to the formation of a blood clot.
- Enzyme Activation: Calcium ions act as cofactors for many enzymes, regulating their activity and playing a role in various metabolic pathways.
- Cell Signaling: Calcium ions participate in intracellular signaling pathways, acting as second messengers that relay signals from the cell surface to the interior of the cell.
Calcium's Applications: From Construction to Medicine
Calcium's versatility extends to a wide range of applications:
- Construction Industry: Calcium compounds like limestone (calcium carbonate, CaCO₃) are extensively used in the construction industry as building materials and in cement production.
- Metallurgy: Calcium is used as an alloying agent in various metal alloys, improving their properties.
- Agriculture: Calcium is an essential nutrient for plants, and calcium-containing fertilizers are used to improve soil fertility.
- Medicine: Calcium supplements are widely used to prevent and treat calcium deficiency, osteoporosis, and other health conditions. Calcium salts are also used in various pharmaceutical preparations.
- Food Industry: Calcium compounds are added to food products as nutritional supplements and food additives.
Conclusion: Calcium – An Indispensable Metallic Element
In summary, calcium’s physical and chemical properties definitively place it within the category of metals. Its high electrical and thermal conductivity, malleability (to a degree), ductility (again, to a degree), low ionization energy, low electronegativity, reactivity with water and acids, and formation of ionic compounds all align perfectly with the defining characteristics of metals. Moreover, its crucial biological roles and diverse applications highlight its indispensable nature in various aspects of life and technology. From the strength of our bones to the function of our nerves, and from building materials to medical treatments, calcium's impact is undeniable. Its metallic nature is fundamental to its diverse properties and vital functions.
Latest Posts
Latest Posts
-
What Is 2 3 Of 100
Apr 05, 2025
-
What State Of Matter Is Lightning
Apr 05, 2025
-
Flood Waters Moving Soil From One Location To Another
Apr 05, 2025
-
What Is 9 In Fraction Form
Apr 05, 2025
-
Is 23 A Prime Or Composite
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Calcium Is A Metal Nonmetal Or Metalloid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
