Choose The Substance With The Highest Surface Tension
Kalali
Apr 05, 2025 · 6 min read
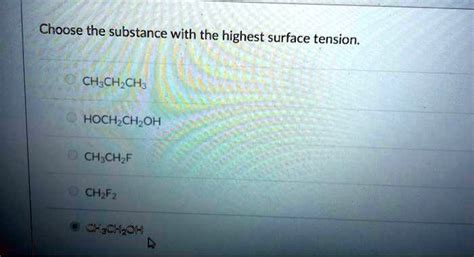
Table of Contents
Choose the Substance with the Highest Surface Tension: A Deep Dive
Surface tension, a captivating phenomenon in the world of physics and chemistry, refers to the tendency of liquid surfaces to shrink into the minimum surface area possible. This intriguing property arises from the cohesive forces between liquid molecules. Understanding and identifying substances with high surface tension is crucial across various scientific disciplines and industrial applications. This article will delve into the intricacies of surface tension, exploring the factors that influence it and ultimately identifying substances known for their exceptionally high surface tension.
Understanding Surface Tension: A Molecular Perspective
At the heart of surface tension lies the interplay of intermolecular forces. Liquid molecules within the bulk experience attractive forces from all surrounding molecules, resulting in a net force of approximately zero. However, molecules at the surface experience a net inward force. This is because they are only surrounded by other liquid molecules on one side and, typically, air or another less dense phase on the other. This imbalance of forces pulls the surface molecules inwards, minimizing the surface area and creating a sort of "skin" on the liquid's surface.
This "skin" is responsible for several observable phenomena, such as the formation of droplets, the ability of certain insects to walk on water, and the capillary action that draws water upwards in narrow tubes. The stronger the intermolecular forces, the greater the surface tension.
Factors Affecting Surface Tension
Several factors significantly influence the surface tension of a liquid:
-
Temperature: As temperature increases, the kinetic energy of the molecules also increases. This enhanced molecular motion disrupts the intermolecular forces, leading to a decrease in surface tension. Generally, surface tension decreases with increasing temperature.
-
Intermolecular Forces: The strength of intermolecular forces plays a crucial role. Stronger forces like hydrogen bonding (present in water) lead to higher surface tension, while weaker forces like van der Waals forces result in lower surface tension. The presence of polar groups in a molecule often contributes to stronger intermolecular interactions and thus higher surface tension.
-
Presence of Impurities: The addition of impurities, such as surfactants or detergents, can significantly alter the surface tension. Surfactants, with their amphiphilic nature (having both hydrophilic and hydrophobic parts), disrupt the cohesive forces between liquid molecules, effectively lowering the surface tension.
-
Concentration: The concentration of a solute can influence surface tension. In many cases, increasing the concentration of a solute initially reduces the surface tension until reaching a saturation point.
Measuring Surface Tension: Techniques and Methods
Several techniques exist for accurately measuring surface tension. These methods provide quantitative data crucial for scientific investigations and industrial quality control:
-
Du Noüy Ring Method: This widely used method involves carefully lifting a platinum ring from the liquid surface. The force required to detach the ring is directly proportional to the surface tension.
-
Wilhelmy Plate Method: A thin, vertical plate (often made of platinum) is partially immersed in the liquid. The force required to pull the plate upwards is measured and used to calculate the surface tension.
-
Pendant Drop Method: This method involves analyzing the shape of a liquid drop hanging from a capillary tube. The shape of the drop is related to the surface tension and can be analyzed using image analysis techniques.
-
Capillary Rise Method: The height to which a liquid rises in a narrow capillary tube is related to the surface tension, density, and contact angle. This method is particularly suitable for liquids with low viscosity.
Each technique offers advantages and disadvantages, with the choice often depending on the specific liquid, desired accuracy, and available resources.
Identifying Substances with High Surface Tension: A Comparative Analysis
While many substances exhibit surface tension, certain liquids stand out due to their exceptionally high values. This high surface tension is often a direct consequence of strong intermolecular forces, primarily hydrogen bonding.
Let's compare a few key substances:
-
Water (H₂O): Water consistently ranks high in terms of surface tension due to its extensive hydrogen bonding network. This strong intermolecular attraction results in a remarkably high surface tension at room temperature, explaining many of water's unique properties.
-
Mercury (Hg): Despite its metallic nature, mercury exhibits exceptionally high surface tension, surpassing even water. This is primarily attributed to the strong metallic bonds within the liquid mercury. However, mercury is highly toxic, and its handling requires significant safety precautions.
-
Glycerol (C₃H₈O₃): Glycerol, a viscous liquid with three hydroxyl groups, also exhibits relatively high surface tension due to the presence of multiple hydrogen bonding sites. It’s often used in various applications, leveraging its high surface tension properties.
Beyond the Usual Suspects: Exploring Other High Surface Tension Liquids
Several other less common substances demonstrate surprisingly high surface tension:
-
Certain molten salts: Some molten salts, particularly those with strong ionic interactions, can exhibit high surface tension. The specific value depends heavily on the composition and temperature.
-
Some organic liquids with strong hydrogen bonding: Certain organic liquids containing multiple hydroxyl or amine groups can possess substantial surface tension. However, their precise values vary significantly based on the molecular structure.
-
Liquid crystals: Some liquid crystals, due to their unique molecular arrangements, display high surface tension. The value varies significantly depending on the type and temperature of the liquid crystal.
It's crucial to note that the precise surface tension of a substance is highly sensitive to temperature and purity. Thus, comparing values requires careful consideration of these factors.
Applications of High Surface Tension Substances
The high surface tension of certain liquids finds numerous applications in various fields:
-
Coatings and Films: Liquids with high surface tension can form uniform, thin coatings and films, making them ideal for applications such as protective coatings, paints, and inks.
-
Microfluidics: In microfluidic devices, the high surface tension of liquids helps to control and manipulate fluid flow in tiny channels.
-
Printing: High surface tension liquids are essential in inkjet printing, ensuring consistent droplet formation and preventing clogging.
-
Wetting and Adhesives: The ability of a liquid to wet a surface depends, in part, on its surface tension. High surface tension can be advantageous in specific adhesive applications.
-
Industrial Processes: High surface tension plays a role in numerous industrial processes, such as metal refining and crystal growth, where careful control over surface properties is crucial.
Conclusion: The Significance of Surface Tension
Understanding and manipulating surface tension is a cornerstone of numerous scientific disciplines and industrial applications. While water is frequently cited for its relatively high surface tension, many other substances, including mercury and certain organic liquids, exhibit even greater values. The choice of the "highest" surface tension substance ultimately depends on the specific conditions and context, requiring careful consideration of temperature, purity, and the measurement techniques used. The exploration and application of high surface tension liquids will undoubtedly continue to drive innovation in various fields. Further research continues to unveil new substances and applications that exploit this fascinating property of matter. The seemingly simple concept of surface tension holds a significant complexity that continues to unravel and offer exciting possibilities for future advancements.
Latest Posts
Latest Posts
-
5 Oz Is How Many Cups
Apr 06, 2025
-
Dogs Belong To The Order Felidae
Apr 06, 2025
-
Do You Need Insulation Wrapped Around Your Fractional Distillation Apparatus
Apr 06, 2025
-
Convert 120 Degrees Celsius To Fahrenheit
Apr 06, 2025
-
Venn Diagram Of Photosynthesis And Respiration
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Choose The Substance With The Highest Surface Tension . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
