Does Oxygen Gain Or Lose Electrons
Kalali
Apr 02, 2025 · 4 min read
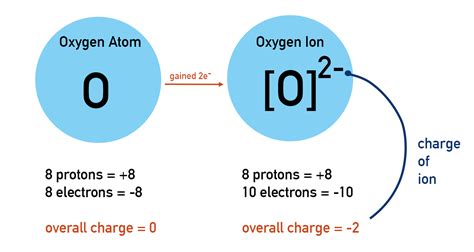
Table of Contents
Does Oxygen Gain or Lose Electrons? Understanding Oxidation and Reduction
Oxygen's role in chemical reactions is fundamentally linked to its electron behavior. The question, "Does oxygen gain or lose electrons?" is not only crucial for understanding basic chemistry but also for grasping complex processes like respiration, combustion, and corrosion. The simple answer is that oxygen almost always gains electrons in chemical reactions. This behavior is a cornerstone of oxidation and reduction (redox) reactions. Let's delve deeper into this fundamental concept.
Understanding Electron Configurations and Electronegativity
To understand why oxygen gains electrons, we need to consider its electron configuration and electronegativity.
Oxygen's Electron Configuration
Oxygen's atomic number is 8, meaning it has 8 protons and 8 electrons in its neutral state. Its electron configuration is 1s²2s²2p⁴. The outermost shell (valence shell) contains 6 electrons. Atoms strive for stability, often achieving this by having a full outermost shell, typically with 8 electrons (the octet rule). Oxygen is two electrons short of a full octet. This electron deficiency drives its chemical behavior.
Electronegativity: Oxygen's Strong Attraction
Electronegativity is a measure of an atom's ability to attract electrons towards itself in a chemical bond. Oxygen has a high electronegativity (3.44 on the Pauling scale), second only to fluorine. This high electronegativity signifies its strong pull on electrons shared in a chemical bond. Consequently, when oxygen interacts with other atoms, it tends to attract electrons more strongly, resulting in electron gain.
Oxidation and Reduction: The Core of Oxygen's Electron Behavior
Oxygen's electron-gaining tendency is central to the concepts of oxidation and reduction, which are always coupled reactions.
Oxidation: Loss of Electrons
Oxidation is defined as the loss of electrons by an atom, molecule, or ion. The substance that loses electrons is said to be oxidized. During oxidation, the oxidation state (or oxidation number) of the atom increases.
Reduction: Gain of Electrons
Reduction is the gain of electrons. The substance that gains electrons is said to be reduced. During reduction, the oxidation state of the atom decreases.
Redox Reactions: A Coupled Process
Oxidation and reduction always occur simultaneously. One substance is oxidized while another is reduced. This simultaneous process is called a redox reaction. In a redox reaction, electrons are transferred from one substance (the reducing agent) to another (the oxidizing agent).
Oxygen as an Oxidizing Agent
Oxygen's high electronegativity makes it a powerful oxidizing agent. This means it readily accepts electrons from other atoms or molecules, causing them to be oxidized. In the process, oxygen itself is reduced.
Examples of Oxygen's Role as an Oxidizing Agent
Let's illustrate with some common examples:
-
Combustion: When a substance burns in air, it reacts with oxygen. The substance loses electrons (is oxidized), and oxygen gains electrons (is reduced). For instance, the combustion of methane (CH₄) can be represented as:
CH₄ + 2O₂ → CO₂ + 2H₂O
In this reaction, carbon in methane goes from an oxidation state of -4 to +4 (oxidation), while oxygen goes from 0 to -2 (reduction).
-
Respiration: Cellular respiration, the process by which living organisms obtain energy, involves the oxidation of glucose (C₆H₁₂O₆) by oxygen. Glucose is oxidized, releasing energy, while oxygen is reduced to form water:
C₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O
-
Corrosion: Rusting of iron is another example of oxygen's oxidizing power. Iron reacts with oxygen and water to form iron oxide (rust):
4Fe + 3O₂ + 6H₂O → 4Fe(OH)₃
Iron is oxidized, and oxygen is reduced.
-
Formation of Oxides: Many metals react with oxygen to form metal oxides. For example, the formation of magnesium oxide:
2Mg + O₂ → 2MgO
Magnesium loses electrons (oxidation), while oxygen gains electrons (reduction).
Exceptions: Peroxides and Superoxides
While oxygen typically gains electrons, there are exceptions involving the formation of peroxides and superoxides.
Peroxides
In peroxides (e.g., hydrogen peroxide, H₂O₂), oxygen has an oxidation state of -1. This means each oxygen atom gains only one electron, not two, compared to its usual -2 state in most oxides. This is a less common oxidation state for oxygen.
Superoxides
Superoxides (e.g., potassium superoxide, KO₂) contain oxygen with an oxidation state of -1/2. In this unusual case, oxygen exists as the superoxide anion (O₂⁻), an oxygen molecule with an extra electron.
Conclusion: Oxygen's Predominant Electron-Gaining Behavior
In summary, oxygen's high electronegativity and its electron configuration strongly favor electron gain. While exceptions exist in peroxides and superoxides, the vast majority of oxygen's chemical reactions involve its function as a powerful oxidizing agent, gaining electrons and undergoing reduction. Understanding this fundamental behavior is essential for comprehending a wide array of chemical and biological processes, from combustion and respiration to corrosion and the formation of oxides. The ability of oxygen to readily accept electrons underpins its crucial role in numerous natural phenomena and industrial applications. This electron-grabbing tendency, while seemingly simple, is the key to unlocking a deep understanding of redox chemistry and its significant impact on our world.
Latest Posts
Latest Posts
-
1 73 M In Inches And Feet
Apr 03, 2025
-
How Many Hours Is 125 Minutes
Apr 03, 2025
-
4 To The Power Of 12
Apr 03, 2025
-
What Are The Disadvantages Of Sexual Reproduction
Apr 03, 2025
-
How Many Inches Is 210 Cm
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Does Oxygen Gain Or Lose Electrons . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
