Is Evaporation Exothermic Or Endothermic Process
Kalali
Apr 06, 2025 · 5 min read
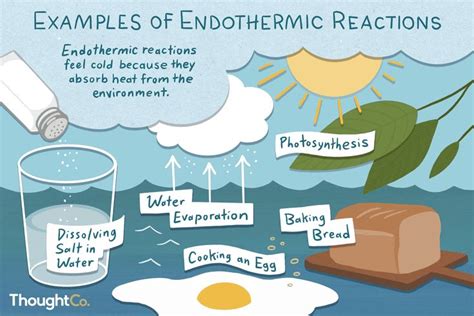
Table of Contents
Is Evaporation an Exothermic or Endothermic Process? A Deep Dive
Evaporation, the transformation of a liquid into a gas, is a fundamental process in nature and crucial in various applications. Understanding its thermodynamic nature – whether it's exothermic or endothermic – is key to comprehending its behavior and impact. This detailed exploration will dissect the intricacies of evaporation, examining its energy requirements, molecular mechanisms, and practical implications.
Understanding Exothermic and Endothermic Processes
Before delving into the specifics of evaporation, let's establish a clear understanding of exothermic and endothermic processes. These terms describe the energy exchange between a system and its surroundings during a physical or chemical change.
-
Exothermic Processes: These processes release energy into their surroundings. The system's energy decreases, and the surroundings' energy increases. This energy release often manifests as heat, leading to a temperature increase in the surroundings. Examples include combustion and the neutralization of acids and bases.
-
Endothermic Processes: These processes absorb energy from their surroundings. The system's energy increases, and the surroundings' energy decreases. This energy absorption often results in a temperature decrease in the surroundings. Examples include melting ice and photosynthesis.
The Thermodynamics of Evaporation: An Endothermic Process
Evaporation is unequivocally an endothermic process. This means it requires energy input to occur. The energy is absorbed from the surroundings, leading to a cooling effect. This energy is primarily used to overcome the intermolecular forces holding the liquid molecules together.
The Role of Intermolecular Forces
Liquids are characterized by relatively strong intermolecular forces, such as hydrogen bonds, dipole-dipole interactions, and van der Waals forces. These forces attract molecules to each other, keeping them relatively close together. For a molecule to escape the liquid phase and transition to the gaseous phase (evaporation), it needs sufficient kinetic energy to overcome these attractive forces.
Kinetic Energy and Molecular Escape
The kinetic energy of molecules is directly related to their temperature. At any given temperature, molecules within a liquid possess a distribution of kinetic energies. Some molecules possess higher kinetic energies than others. Molecules with sufficiently high kinetic energy can overcome the intermolecular forces and escape into the gaseous phase. This is the essence of evaporation.
Energy Absorption and Cooling Effect
The energy required to overcome the intermolecular forces is absorbed from the surrounding environment. This absorption of energy leads to a decrease in the temperature of the remaining liquid and the surrounding environment. This is why sweating is an effective cooling mechanism; as sweat evaporates, it absorbs heat from your body, lowering your temperature.
Factors Affecting Evaporation Rate
Several factors influence the rate of evaporation, all of which relate to the availability of energy and the ease with which molecules can escape the liquid surface:
1. Temperature:
Higher temperatures lead to a higher rate of evaporation. A higher temperature means a larger proportion of molecules possess the kinetic energy needed to overcome intermolecular forces.
2. Surface Area:
A larger surface area increases the rate of evaporation. A larger surface provides more opportunities for molecules to escape the liquid phase. This is why spreading out a liquid will increase its evaporation rate.
3. Humidity:
Lower humidity promotes faster evaporation. Humidity refers to the amount of water vapor already present in the air. If the air is already saturated with water vapor, there's less room for additional water molecules to escape into the gaseous phase, slowing down evaporation.
4. Air Movement (Wind):
Increased air movement accelerates evaporation. Wind removes water vapor molecules from the vicinity of the liquid surface, creating a lower concentration of water vapor in the air above the liquid. This gradient in concentration encourages more water molecules to escape into the gaseous phase.
5. Nature of the Liquid:
The type of liquid significantly affects its evaporation rate. Liquids with weaker intermolecular forces evaporate more readily than those with stronger intermolecular forces. For example, ethanol evaporates faster than water because its intermolecular forces are weaker.
Practical Applications of Evaporation
The endothermic nature of evaporation has widespread practical applications:
-
Cooling Systems: Evaporation is a fundamental principle behind many cooling systems, including sweat-based cooling in humans and animals, refrigeration systems, and evaporative coolers.
-
Drying Processes: Industrial and household drying processes rely on evaporation to remove water from various materials, such as clothes, food, and industrial products.
-
Distillation: Distillation utilizes evaporation and condensation to separate components of a liquid mixture based on their boiling points.
-
Water Cycle: Evaporation plays a crucial role in the Earth's water cycle, driving the transfer of water from the Earth's surface to the atmosphere.
-
Climate Regulation: Evaporation and subsequent condensation processes are key components of the Earth's climate system, influencing temperature and precipitation patterns.
Misconceptions about Evaporation
There are some common misconceptions regarding evaporation:
-
Evaporation is not simply boiling: Boiling occurs at a specific temperature (the boiling point) where vapor pressure equals atmospheric pressure. Evaporation, on the other hand, occurs at any temperature below the boiling point.
-
Evaporation doesn't require a specific temperature: Unlike boiling, evaporation doesn't require reaching a specific temperature threshold. It happens continuously at temperatures below the boiling point.
Conclusion: Evaporation – An Endothermic Process with Far-Reaching Implications
In conclusion, evaporation is definitively an endothermic process, absorbing energy from its surroundings to overcome intermolecular forces and facilitate the transition of liquid molecules into the gaseous phase. This energy absorption leads to a cooling effect, a principle harnessed in various applications across diverse fields. Understanding the endothermic nature of evaporation, along with the factors influencing its rate, provides crucial insights into natural processes and technological innovations, highlighting its fundamental importance in various aspects of science and technology. From cooling mechanisms to industrial drying, the impact of evaporation is undeniable and deeply rooted in its thermodynamic nature. The ongoing research in areas like advanced materials science and atmospheric physics continues to refine our understanding of this fascinating process and its multifaceted consequences.
Latest Posts
Latest Posts
-
Fungi Have Cell Walls Made Of Peptidoglycan
Apr 06, 2025
-
5 Feet 5 Inches Is How Many Centimeters
Apr 06, 2025
-
Reacts With Water Physical Or Chemical Property
Apr 06, 2025
-
Distance Between Two Points In Polar Coordinates
Apr 06, 2025
-
How Long Is 70 Cm In Inches
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Is Evaporation Exothermic Or Endothermic Process . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.