Is Grain Alcohol A Pure Substance
Kalali
Apr 02, 2025 · 5 min read
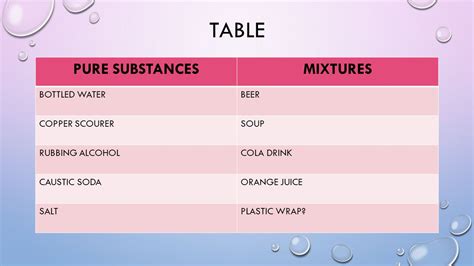
Table of Contents
Is Grain Alcohol a Pure Substance? A Deep Dive into Ethanol Production and Purity
The question of whether grain alcohol is a pure substance is more nuanced than a simple yes or no. While the goal of grain alcohol production is to create pure ethanol (C₂H₅OH), the reality is often more complex. Understanding this requires exploring the ethanol production process, the definition of a pure substance, and the potential contaminants present in various types of grain alcohol.
Understanding the Definition of a Pure Substance
In chemistry, a pure substance is defined as a material that has a constant composition (it's a single element or a single compound) and distinct chemical properties. A pure substance cannot be separated into other substances through physical means like filtration or distillation. This is crucial to understanding the purity of grain alcohol. Pure ethanol, in theory, would meet this definition.
The Grain Alcohol Production Process: From Grain to Ethanol
The production of grain alcohol, also known as ethanol, involves several key steps:
1. Fermentation: Converting Sugars to Ethanol
The process begins with grains like corn, barley, wheat, or rye. These grains contain starches, which need to be broken down into simpler sugars before fermentation can occur. This is achieved through a process called saccharification, often involving enzymes. The resulting sugar solution then undergoes fermentation, where yeast consumes the sugars and converts them into ethanol and carbon dioxide. This is a biological process, inherently less precise than chemical synthesis.
2. Distillation: Separating Ethanol from Other Compounds
The fermented mixture, known as "wash," contains ethanol, water, and various other compounds produced by the yeast and present in the original grain. Distillation is the crucial step that separates the ethanol from these other components. This process relies on the differing boiling points of the components: ethanol boils at 78.4°C, while water boils at 100°C. Through multiple distillation cycles, the concentration of ethanol can be significantly increased.
3. Purification: Reaching High Ethanol Concentrations
Even after multiple distillations, the resulting alcohol isn't perfectly pure. It still contains traces of other alcohols (like methanol, propanol), esters, aldehydes, and congeners – compounds that contribute to the flavor and aroma profile of different alcoholic beverages. The level of these congeners varies significantly depending on the grain source, fermentation process, and the number of distillations.
The Impact of Different Distillation Methods on Purity
The purity of grain alcohol is heavily influenced by the distillation method employed.
Simple Distillation: This method offers a relatively low level of purity, leaving behind a significant amount of congeners. This is typical for many spirits that aim for a specific flavor profile, rather than pure ethanol.
Fractional Distillation: This more advanced technique allows for a higher degree of separation, resulting in a higher concentration of ethanol. Multiple distillation columns increase the efficiency of separation, leading to greater purity.
Rectification: This process, often employed in the production of neutral grain spirits (NGS), utilizes highly efficient distillation columns to achieve extremely high ethanol concentrations – often over 95%. NGS serves as a base for various spirits, and its purity is crucial.
What Contaminants Can Be Found in Grain Alcohol?
Even in highly purified grain alcohol, trace amounts of various compounds remain. These include:
-
Methanol (CH₃OH): A highly toxic alcohol, methanol is a significant concern in improperly produced grain alcohol. Its presence is a strong indicator of impure product.
-
Higher alcohols (propanol, butanol): These contribute to the flavor profile of alcoholic beverages, but excessive amounts can be undesirable.
-
Aldehydes and esters: These are formed during fermentation and distillation, contributing to the taste and aroma, but their presence implies the product is not pure ethanol.
-
Fusel oils: A mixture of higher alcohols with unpleasant tastes and odors, fusel oils are a common byproduct of fermentation.
-
Acetaldehyde: A byproduct of ethanol oxidation, this can result in hangovers and other unpleasant effects.
The presence and concentration of these contaminants determine the purity of the grain alcohol. The absence of these contaminants is a hallmark of a genuinely pure substance.
The Role of Denaturants in Grain Alcohol
Grain alcohol intended for industrial or non-beverage use is often denatured, meaning substances are added to make it unfit for consumption. Common denaturants include methanol, isopropyl alcohol, and bittering agents. This renders the alcohol poisonous, preventing misuse and abuse. Denatured alcohol is explicitly not a pure substance.
Determining the Purity of Grain Alcohol: Analytical Methods
Several analytical methods can accurately determine the purity of grain alcohol:
-
Gas chromatography (GC): This technique separates the different components of a mixture based on their boiling points, allowing for precise quantification of each component, including ethanol and contaminants.
-
High-performance liquid chromatography (HPLC): Similar to GC, HPLC offers precise separation and quantification of compounds in a mixture.
-
Spectroscopy: Various spectroscopic techniques, such as near-infrared (NIR) spectroscopy, can rapidly analyze the composition of grain alcohol, providing a quick assessment of its purity.
These analytical methods are crucial for ensuring the quality and safety of grain alcohol intended for various applications.
Grain Alcohol: Pure Ethanol or a Complex Mixture?
In conclusion, the answer to whether grain alcohol is a pure substance is complex. While the goal of production is to obtain pure ethanol, the reality is that most grain alcohol contains trace amounts of other compounds, even after multiple distillations and purification steps. The extent of these impurities determines its level of purity. Highly purified grain alcohol, like neutral grain spirits (NGS), approaches pure ethanol, but it never truly reaches the definition of a pure chemical substance because of the presence of traces of other compounds. Only exceptionally pure, laboratory-grade ethanol produced through advanced chemical synthesis would meet the strict definition of a pure substance. Therefore, considering the complexities of the production process and the ever-present trace impurities, it's more accurate to describe grain alcohol as a predominantly ethanol mixture, rather than a pure substance. The purity level depends entirely on the production method and the intended use.
Latest Posts
Latest Posts
-
1 73 M In Inches And Feet
Apr 03, 2025
-
How Many Hours Is 125 Minutes
Apr 03, 2025
-
4 To The Power Of 12
Apr 03, 2025
-
What Are The Disadvantages Of Sexual Reproduction
Apr 03, 2025
-
How Many Inches Is 210 Cm
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is Grain Alcohol A Pure Substance . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
