Number Of Energy Levels In Oxygen
Kalali
Apr 01, 2025 · 5 min read
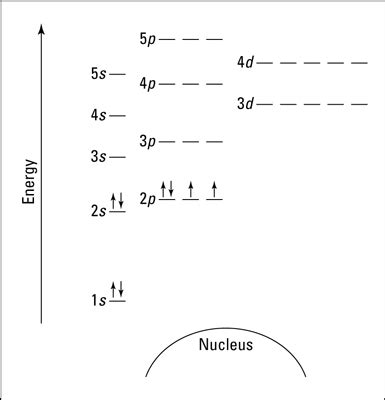
Table of Contents
Delving into the Electronic Structure of Oxygen: Understanding its Energy Levels
Oxygen, a life-sustaining element, boasts a fascinating electronic structure that dictates its chemical reactivity and diverse roles in various systems. Understanding the number of energy levels in oxygen requires exploring its atomic structure, electron configuration, and the principles of quantum mechanics that govern electron behavior within an atom. This in-depth exploration will go beyond a simple answer, providing a comprehensive understanding of oxygen's electronic structure and its implications.
Oxygen's Atomic Structure: Setting the Stage
Oxygen, with an atomic number of 8, possesses eight protons in its nucleus and, in its neutral state, eight electrons orbiting the nucleus. These electrons aren't randomly distributed; they occupy specific energy levels, or shells, characterized by their principal quantum number (n). These shells are further subdivided into subshells, designated by the azimuthal quantum number (l), which determines the shape of the electron orbitals.
Principal Energy Levels and Subshells
The principal energy levels (n = 1, 2, 3, etc.) represent the average distance of electrons from the nucleus. Lower values of 'n' correspond to electrons closer to the nucleus and lower energy. Within each principal energy level, subshells exist, each possessing a specific energy and shape:
-
n=1 (K shell): This shell contains only one subshell: the 1s subshell, which can hold a maximum of two electrons. These electrons experience the strongest attraction to the nucleus.
-
n=2 (L shell): This shell contains two subshells: the 2s and 2p subshells. The 2s subshell can hold up to two electrons, while the 2p subshell can accommodate up to six electrons (three orbitals, each holding two electrons).
-
n=3 (M shell) and beyond: While higher energy levels exist theoretically, they are largely unoccupied in ground-state oxygen.
Oxygen's Electron Configuration and Energy Level Occupancy
The electron configuration of an atom describes how its electrons are distributed among the various energy levels and subshells. Oxygen's electron configuration is 1s²2s²2p⁴. This indicates:
- 1s²: Two electrons occupy the 1s subshell (lowest energy).
- 2s²: Two electrons occupy the 2s subshell.
- 2p⁴: Four electrons occupy the 2p subshell. Remember, the 2p subshell has three orbitals (px, py, pz), each capable of holding two electrons. In oxygen, these four electrons are distributed according to Hund's rule, which states that electrons will individually occupy each orbital within a subshell before pairing up. This leads to two singly occupied orbitals and one doubly occupied orbital in the 2p subshell.
The Significance of the 2p Subshell in Oxygen's Reactivity
The partially filled 2p subshell is crucial in understanding oxygen's chemical behavior. This incompletely filled subshell signifies that oxygen has a high tendency to gain two electrons to achieve a stable octet configuration, characteristic of the noble gases. This electron-seeking behavior makes oxygen highly reactive and explains its role in various chemical reactions, including combustion and respiration.
Energy Level Differences and Transitions
The electrons within different energy levels and subshells possess differing energies. When an electron absorbs energy (e.g., from light or heat), it can transition to a higher energy level. This is an excited state. Conversely, when an electron falls from a higher energy level to a lower one, it emits energy, often in the form of light (e.g., the characteristic spectral lines of oxygen). The energy difference between energy levels determines the frequency and wavelength of the emitted or absorbed light.
Beyond the Simple Model: Advanced Considerations
The simple model presented above provides a basic understanding. A more accurate depiction requires considerations like:
-
Electron-Electron Repulsion: The model assumes electrons are independent particles, ignoring the significant repulsion between them. This repulsion influences the precise energy levels and orbital shapes.
-
Spin-Orbit Coupling: The interaction between the electron's spin and its orbital angular momentum slightly alters the energy levels, especially in heavier atoms.
-
Quantum Mechanical Calculations: Accurate calculations of energy levels require sophisticated computational techniques, using methods like Hartree-Fock or Density Functional Theory (DFT), to account for the complex interactions within the atom. These calculations provide a more precise understanding of the energy differences between orbitals and the impact of various quantum effects.
Practical Implications and Applications
Understanding oxygen's energy levels is crucial across multiple scientific and engineering disciplines:
-
Chemistry: Predicting reaction rates, bonding characteristics, and the stability of oxygen-containing compounds.
-
Materials Science: Designing materials with specific properties by manipulating oxygen's electronic structure through doping or other techniques.
-
Physics: Understanding spectral lines in astronomical observations to analyze the composition of stars and other celestial objects.
-
Biochemistry: Explaining oxygen's role in respiration and its interaction with biological molecules. The energy transfer during oxidation processes depends on the electron's energy levels within oxygen and other molecules.
-
Environmental Science: Monitoring atmospheric oxygen levels and assessing the impact of pollution. The electronic structure impacts oxygen's ability to react with pollutants.
Conclusion: A Deeper Dive into Oxygen's Electronic Complexity
While we often simplify oxygen's structure to a basic model of energy levels, the reality is far more complex. Understanding its electron configuration, the subtle nuances of electron-electron interactions, and the advanced quantum mechanical descriptions reveals a rich tapestry of energy levels and their profound impact on oxygen's properties and its essential roles in various natural processes. This detailed exploration reveals the importance of going beyond basic models to appreciate the intricacies of atomic structure and its far-reaching consequences. The more we understand the intricacies of oxygen's electronic structure, the better equipped we are to harness its properties for scientific advancements and to better understand the world around us. The future of research in this area undoubtedly holds exciting advancements in our understanding of this ubiquitous and vital element.
Latest Posts
Latest Posts
-
How Many Inches Is 42 Centimeters
Apr 03, 2025
-
What Does A Turtle Look Like Without The Shell
Apr 03, 2025
-
Cuanto Es 31 Grados Fahrenheit En Centigrados
Apr 03, 2025
-
Which Linear Equation Shows A Proportional Relationship
Apr 03, 2025
-
Why Are Cathode Ray Tubes Connected To A Vacuum Pump
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Number Of Energy Levels In Oxygen . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
