Toasting Bread Physical Or Chemical Change
Kalali
Apr 02, 2025 · 6 min read
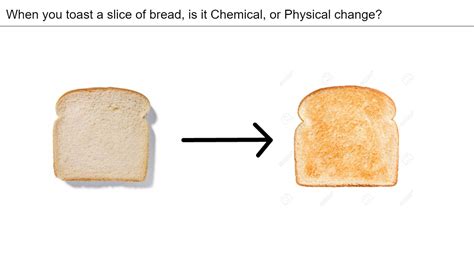
Table of Contents
Toasting Bread: A Deep Dive into Physical and Chemical Changes
Toasting bread, a seemingly simple act, offers a fascinating glimpse into the world of chemistry and physics. While it may appear to be a mere alteration of texture and color, the process involves a complex interplay of physical and chemical changes that transform a slice of bread into a crispy, golden-brown delight. This article delves deep into the science behind toasting bread, examining the physical transformations, the chemical reactions involved, and the impact of different toasting methods.
The Physical Changes: More Than Meets the Eye
The most readily observable changes during toasting are physical. These transformations don't involve altering the chemical composition of the bread itself, but rather its structure and properties. Let's explore these key physical changes:
1. Water Evaporation: The Foundation of Crispness
Bread, even after baking, contains a significant amount of water. When exposed to the high heat of a toaster, this water begins to evaporate. This is a physical change, as the water is simply changing state from liquid to gas, not undergoing any alteration in its chemical makeup. The removal of water is crucial for achieving that characteristic crisp texture of toast. The drier the bread becomes, the crispier it will be. Over-toasting, however, can lead to burning, as we will explore later.
2. Starch Gelatinization and Retrogradation: A Complex Dance
Bread primarily consists of starch, a complex carbohydrate. During baking, the starch granules absorb water and swell, a process known as gelatinization. This process contributes significantly to the texture of the bread. However, toasting introduces a new dynamic – retrogradation. As the bread dries during toasting, the starch molecules begin to realign and recrystallize, resulting in a firmer, more rigid structure. This retrogradation contributes to the crispness and crunch of toast.
3. Structural Changes: From Soft to Crisp
The initial soft structure of bread is significantly altered during toasting. The heat causes the bread's internal structure to undergo significant changes. The evaporation of water and the retrogradation of starch contribute to the reduction in volume and increase in rigidity. Microscopic structures within the bread are altered, resulting in the characteristic crisp and firm texture.
The Chemical Changes: The Maillard Reaction and Beyond
While the physical changes are readily apparent, the chemical transformations during toasting are equally important and often more subtle. These changes dramatically impact the flavor, aroma, and color of the toast.
1. The Maillard Reaction: The Chemistry of Browning and Flavor
The most significant chemical change responsible for the brown color and characteristic aroma of toast is the Maillard reaction. This complex series of reactions occurs between amino acids (the building blocks of proteins) and reducing sugars (like glucose) when heated. The Maillard reaction isn't a single reaction, but rather a cascade of reactions producing hundreds of different compounds, many of which contribute to the complex flavor profile of toasted bread.
The Maillard reaction is crucial for:
- Color Development: The browning observed during toasting is a direct result of the formation of melanoidins, dark-colored polymers produced during the Maillard reaction.
- Flavor Enhancement: The reaction produces a vast array of volatile compounds responsible for the unique aroma and taste of toast, including aldehydes, ketones, and pyrazines. These compounds contribute to the nutty, savory, and slightly sweet notes we associate with toast.
- Nutritional Changes: While some nutrients may be lost during toasting due to heat degradation, the Maillard reaction can also create new compounds with potential health benefits, although research in this area is ongoing.
2. Caramelization: Sweetening the Deal
In addition to the Maillard reaction, caramelization plays a role in the browning and flavor development of toast. Caramelization is the process of heating sugars to high temperatures, resulting in the formation of caramel. The sugars in the bread undergo caramelization, contributing to the brown color and sweetness of the toast. This process is particularly noticeable in toasting bread with a higher sugar content.
3. Degradation of Nutrients: A Double-Edged Sword
While toasting enhances flavor and aroma, it's crucial to acknowledge that some nutritional losses can occur. High temperatures can degrade certain vitamins and other sensitive nutrients. However, the extent of nutrient loss depends on the toasting time and temperature. Moderate toasting minimizes nutritional losses while still delivering desirable textural and flavor changes.
Different Toasting Methods: Variations on a Theme
The toasting process itself can be modified, leading to variations in the resulting physical and chemical changes.
1. Toasting in a Toaster: The Conventional Approach
The standard toaster provides consistent and even heating, resulting in uniform browning and crispness. The controlled heat facilitates both the physical and chemical changes described above, producing a consistently enjoyable result.
2. Toasting on a Grill or Pan: Achieving Deeper Browning
Grilling or pan-toasting offers greater control over heat and allows for more pronounced browning and potentially deeper caramelization. Direct contact with the hot surface facilitates faster heat transfer, leading to more significant Maillard reaction and potentially a more intensely flavored toast.
3. Toasting in an Oven: An Alternative Approach
Oven toasting allows for the toasting of larger quantities of bread and provides a slightly different heat profile compared to toasters. The slower, more indirect heat can lead to a slightly different texture and flavor profile.
Understanding the Science Behind a Perfect Slice
The seemingly simple act of toasting bread reveals a complex interplay of physical and chemical processes. From the evaporation of water to the intricacies of the Maillard reaction and caramelization, the transformation from a soft slice of bread to a crispy piece of toast is a testament to the power of heat and chemical reactions. By understanding these processes, you can better control the toasting process to achieve your desired level of crispness, color, and flavor. Whether you prefer a lightly toasted slice or a deeply browned and crunchy piece, the science of toasting bread offers a fascinating exploration of everyday chemistry and physics. Experiment with different methods, and discover the perfect toast for your palate!
Further Exploration: Beyond the Basics
This discussion primarily focuses on white bread. However, the toasting process varies slightly depending on the type of bread. Whole-wheat bread, for example, may toast differently due to its higher fiber content and different composition of starches and sugars. The presence of seeds or grains might also impact the toasting process. Furthermore, exploring the impact of different types of bread on the Maillard reaction and caramelization would provide further insight into the nuances of toasting. This could be a subject for future research. The potential impact of antioxidants present in different types of bread on the overall chemical reactions also warrants investigation. Finally, examining the potential for the development of acrylamide, a potential carcinogen formed during high-temperature cooking of starchy foods, and its link to toasting techniques is a crucial area for future study. It would be beneficial to investigate methods to minimize acrylamide formation while still achieving desirable taste and texture.
Latest Posts
Latest Posts
-
800 Minutes Is How Many Hours
Apr 03, 2025
-
How Many Inches Is 108 Centimeters
Apr 03, 2025
-
Dominio Y Rango De Una Funcion
Apr 03, 2025
-
Convert 24 Degrees Celsius To Fahrenheit
Apr 03, 2025
-
From 40 How Many To 186
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Toasting Bread Physical Or Chemical Change . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
