What Is The Boiling Point Of Salty Water
Kalali
Apr 03, 2025 · 5 min read
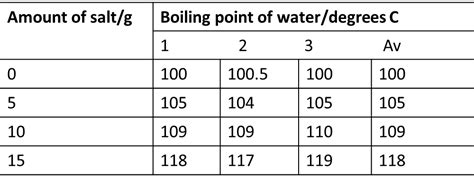
Table of Contents
What is the Boiling Point of Salty Water? A Deep Dive into Colligative Properties
The seemingly simple question, "What is the boiling point of salty water?" opens a fascinating window into the world of chemistry and, more specifically, colligative properties. While pure water boils at 100°C (212°F) at standard atmospheric pressure, adding salt significantly alters this point. This article will explore the science behind this phenomenon, examining the factors that influence the boiling point elevation and the practical implications of this effect.
Understanding Boiling Point Elevation
The boiling point of a liquid is the temperature at which its vapor pressure equals the surrounding atmospheric pressure. When you heat water, its molecules gain kinetic energy, allowing some to overcome the intermolecular forces holding them together and escape as vapor. At the boiling point, this escape rate is high enough to create a substantial vapor pressure that matches the atmospheric pressure, resulting in the vigorous bubbling we associate with boiling.
Adding a solute, like salt (NaCl), to water changes the boiling point. This phenomenon is known as boiling point elevation, a colligative property. Colligative properties depend solely on the number of solute particles present in a solution, not their identity. In other words, it's not the type of salt that matters as much as how much salt is dissolved.
The key mechanism behind boiling point elevation is the disruption of the solvent-solvent interactions. Salt dissolves in water by dissociating into its constituent ions: Na⁺ and Cl⁻. These ions interact with water molecules, effectively reducing the number of water molecules free to escape into the vapor phase. To achieve the same vapor pressure as pure water, the solution needs to reach a higher temperature. This is why salty water boils at a higher temperature than pure water.
Factors Affecting Boiling Point Elevation
Several factors influence the extent to which the boiling point of water is elevated by adding salt:
1. Concentration of the Solute
The most significant factor is the concentration of the salt in the solution. A higher salt concentration leads to a greater number of ions interacting with water molecules, resulting in a more pronounced elevation of the boiling point. This relationship is generally proportional, meaning doubling the salt concentration roughly doubles the boiling point elevation (though this is an approximation, as we'll discuss later).
2. Type of Solute
While the number of particles is the primary determinant, the type of solute also plays a minor role. The extent of dissociation affects the number of particles in solution. For example, NaCl dissociates into two ions (Na⁺ and Cl⁻) per formula unit, while a molecule like glucose (C₆H₁₂O₆) remains undissociated, meaning only one particle per molecule contributes to the colligative effect. A solution of NaCl at a given concentration will exhibit a larger boiling point elevation than a glucose solution of the same concentration.
This is quantified by the van't Hoff factor (i), which represents the effective number of particles a solute dissociates into in solution. For NaCl, the ideal van't Hoff factor is 2. However, in reality, this value can be less than 2 due to ion pairing and other interactions, especially at high concentrations.
3. Atmospheric Pressure
Atmospheric pressure also influences the boiling point. Higher atmospheric pressure requires a higher vapor pressure to achieve boiling, thus raising the boiling point for both pure water and salty water. Conversely, lower atmospheric pressure lowers the boiling point. This is why water boils at a lower temperature at high altitudes where the atmospheric pressure is reduced.
4. Temperature
The effect of temperature on boiling point elevation is indirect and complex. While temperature directly increases the kinetic energy of the molecules, affecting the rate of evaporation, its primary role is in influencing the solubility of the salt. Solubility changes with temperature – the higher the temperature, the more salt, generally, that can dissolve into the water. Increased salt concentration thus leads to a higher boiling point.
Calculating Boiling Point Elevation
The boiling point elevation (ΔTb) can be estimated using the following equation:
ΔTb = Kb * m * i
Where:
- ΔTb is the boiling point elevation (in °C or °F)
- Kb is the ebullioscopic constant for water (0.512 °C/m)
- m is the molality of the solution (moles of solute per kilogram of solvent)
- i is the van't Hoff factor
This equation provides a reasonable approximation, particularly for dilute solutions. However, it becomes less accurate at higher concentrations due to deviations from ideal behavior, including ion pairing and changes in solvent activity. More complex models are needed to accurately predict boiling point elevation in concentrated solutions.
Practical Implications of Boiling Point Elevation
The boiling point elevation of salty water has several practical applications:
-
Cooking: Adding salt to water during cooking increases its boiling point, allowing for slightly faster cooking times. However, the effect is relatively small and often overshadowed by other factors.
-
Chemical Engineering: Boiling point elevation is critical in many industrial processes involving solutions. Understanding and controlling the boiling point is essential for optimizing separation and purification techniques.
-
Desalination: Desalination processes, which remove salt from seawater to produce freshwater, rely on the principles of boiling point elevation. However, in this case, the goal is to reduce the salinity by removing the water through evaporation and condensation.
-
Anti-freeze: While this article focuses on boiling points, the concept directly relates to freezing point depression - another colligative property. The addition of solutes lowers the freezing point of water. This is the fundamental principle behind using antifreeze solutions in cars to prevent freezing in cold weather.
Conclusion
The boiling point of salty water is higher than that of pure water due to the colligative property of boiling point elevation. The extent of this elevation depends on the concentration of salt, the type of solute, atmospheric pressure, and temperature. While a simple equation can provide an estimate, more complex models are needed for accurate predictions, particularly at high concentrations. Understanding this phenomenon is crucial in various scientific and engineering applications, highlighting the importance of colligative properties in solution chemistry. This knowledge allows for better control and optimization of processes involving salt solutions, ranging from simple cooking to complex industrial operations. Further research continually refines our understanding of this important property, improving the accuracy of predictions and expanding its applicability in diverse fields.
Latest Posts
Latest Posts
-
52 Is What Percent Of 80
Apr 04, 2025
-
How Many Seconds In 35 Minutes
Apr 04, 2025
-
What Is 4 As A Fraction
Apr 04, 2025
-
What Percent Is 12 Of 50
Apr 04, 2025
-
Which Discovery Supported The Endosymbiotic Theory
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The Boiling Point Of Salty Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
