When Does The Given Chemical System Reach Dynamic Equilibrium
Kalali
Apr 03, 2025 · 6 min read
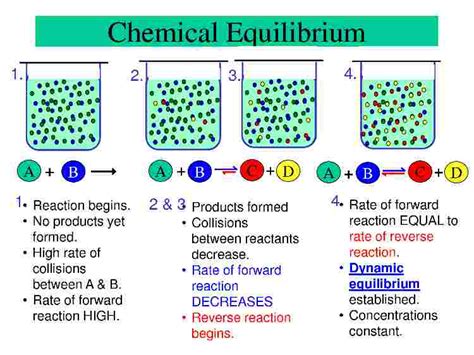
Table of Contents
When Does a Chemical System Reach Dynamic Equilibrium?
Understanding dynamic equilibrium is crucial for comprehending chemical reactions and predicting their behavior. It's a concept that often causes confusion, but with a clear explanation and relatable examples, it becomes much easier to grasp. This article delves deep into the conditions necessary for a chemical system to reach dynamic equilibrium, exploring the factors influencing its attainment and the characteristics that define this state.
What is Dynamic Equilibrium?
Dynamic equilibrium, in the context of reversible chemical reactions, describes a state where the rates of the forward and reverse reactions are equal. This doesn't mean the reaction has stopped; instead, both the forward and reverse reactions continue at the same pace. The concentrations of reactants and products remain constant over time because the rate of formation of products equals the rate of their conversion back into reactants. Imagine a busy highway with cars flowing in both directions – at dynamic equilibrium, the number of cars heading in each direction is the same, even though cars are constantly moving.
Key characteristics of dynamic equilibrium:
- Equal rates: The forward and reverse reaction rates are equal.
- Constant concentrations: The concentrations of reactants and products remain constant over time.
- Reversible reaction: Dynamic equilibrium only occurs in reversible reactions, reactions that can proceed in both the forward and reverse directions.
- Closed system: The system must be closed, meaning no matter can enter or leave. This ensures that the total amount of reactants and products remains constant.
Factors Affecting the Attainment of Dynamic Equilibrium
Several factors influence how quickly, or even if, a chemical system reaches dynamic equilibrium. Understanding these factors is key to controlling and predicting reaction outcomes.
1. Nature of the Reactants and Products
The inherent properties of the reactants and products play a significant role. Some reactions are inherently faster than others due to their activation energy (the energy required to initiate the reaction). Reactions with low activation energies reach equilibrium faster than those with high activation energies. The chemical structure and bonding of the reactants and products also impact the reaction rates. For example, reactions involving simple ionic compounds tend to be faster than those involving complex organic molecules.
2. Temperature
Temperature significantly impacts reaction rates. Increasing the temperature generally increases the kinetic energy of the molecules, leading to more frequent and energetic collisions. This, in turn, increases both the forward and reverse reaction rates. However, the effect on the equilibrium position (the relative amounts of reactants and products at equilibrium) depends on whether the reaction is exothermic (releases heat) or endothermic (absorbs heat). According to Le Chatelier's principle, increasing the temperature of an exothermic reaction shifts the equilibrium towards the reactants, while increasing the temperature of an endothermic reaction shifts the equilibrium towards the products.
3. Concentration
Changing the concentration of reactants or products disrupts the equilibrium. According to Le Chatelier's principle, adding more reactants will shift the equilibrium towards the products, increasing the rate of the forward reaction until a new equilibrium is established. Conversely, adding more products will shift the equilibrium towards the reactants. Similarly, removing reactants or products will also shift the equilibrium to compensate for the loss.
4. Pressure
Pressure affects the equilibrium of reactions involving gases. Increasing the pressure favors the side of the reaction with fewer gas molecules. This is because increasing pressure forces the molecules closer together, making collisions more frequent and favoring the side with less overall volume. Conversely, decreasing the pressure favors the side with more gas molecules.
5. Presence of a Catalyst
A catalyst speeds up both the forward and reverse reactions equally. It does not affect the equilibrium position but significantly accelerates the rate at which equilibrium is reached. Catalysts achieve this by providing an alternative reaction pathway with a lower activation energy.
Determining When Equilibrium is Reached
Experimentally, determining when a system has reached dynamic equilibrium involves monitoring the concentrations of reactants and products over time. When the concentrations remain constant, even though the forward and reverse reactions are still occurring, the system has reached dynamic equilibrium. This can be achieved using various analytical techniques, such as spectrophotometry (measuring light absorption), chromatography (separating components of a mixture), or titration (determining the concentration of a substance).
Graphically, you'll observe a plateau in the concentration-time graphs of both reactants and products. The slopes of the concentration versus time graphs will approach zero as the system approaches equilibrium, indicating that the rate of change in concentration is negligible.
Examples of Dynamic Equilibrium
Several real-world examples illustrate the concept of dynamic equilibrium:
-
Dissolution of a sparingly soluble salt: When a sparingly soluble salt, like silver chloride (AgCl), is added to water, it dissolves to a limited extent, forming silver and chloride ions. Simultaneously, these ions recombine to form the solid salt. At equilibrium, the rate of dissolution equals the rate of precipitation, and the concentrations of the ions remain constant.
-
Phase equilibrium: The evaporation of water in a closed container is an example of dynamic equilibrium. Water molecules continuously escape from the liquid phase into the gaseous phase (evaporation), while simultaneously, water molecules in the gas phase condense back into the liquid phase. At equilibrium, the rates of evaporation and condensation are equal, and the vapor pressure remains constant.
-
Biochemical reactions: Many biochemical reactions in living organisms operate under conditions of dynamic equilibrium. For example, the synthesis and breakdown of ATP (adenosine triphosphate), the energy currency of cells, is a reversible process that maintains a dynamic equilibrium to meet the energy demands of the cell.
Distinguishing Dynamic Equilibrium from Static Equilibrium
It is important to distinguish dynamic equilibrium from static equilibrium. In static equilibrium, there is no change in the system at all – the reaction has essentially stopped. In contrast, in dynamic equilibrium, both the forward and reverse reactions are still occurring at equal rates, resulting in no net change in concentrations. The constant concentrations are a result of continuous, equal and opposite changes.
Applications of Understanding Dynamic Equilibrium
The understanding of dynamic equilibrium has wide-ranging applications in various fields, including:
- Chemical Engineering: Designing and optimizing chemical processes often requires careful consideration of equilibrium conditions to maximize product yield and efficiency.
- Environmental Science: Understanding dynamic equilibrium is critical for analyzing pollution levels and predicting the fate of pollutants in the environment.
- Medicine: Many drug interactions and metabolic processes involve dynamic equilibria that influence drug efficacy and side effects.
- Materials Science: The synthesis and properties of many materials are governed by dynamic equilibria at the molecular level.
Conclusion
Reaching dynamic equilibrium in a chemical system is a complex process influenced by multiple factors. By understanding the role of temperature, concentration, pressure, the nature of reactants and products and the presence of catalysts, we can better control and predict the behavior of chemical reactions. The concept of dynamic equilibrium is fundamental to chemistry and its applications, providing a framework for understanding the intricate interplay of forward and reverse reactions in countless natural and engineered systems. Recognizing that equilibrium is a dynamic state, not a static one, is crucial for grasping the true nature of chemical processes. By monitoring the concentrations of reactants and products over time, we can ascertain when dynamic equilibrium is attained, opening a pathway for understanding and manipulating various chemical systems for technological and scientific advancements.
Latest Posts
Latest Posts
-
Is 29 A Prime Number Or A Composite Number
Apr 04, 2025
-
How Many Valence Electrons Does A Calcium Atom Have
Apr 04, 2025
-
How Many Oz In A Cup Of Butter
Apr 04, 2025
-
How Many Inches In 115 Cm
Apr 04, 2025
-
What Is 140 Out Of 200
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about When Does The Given Chemical System Reach Dynamic Equilibrium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
