Which Products Are The Result Of A Neutralization Reaction
Kalali
Apr 03, 2025 · 6 min read
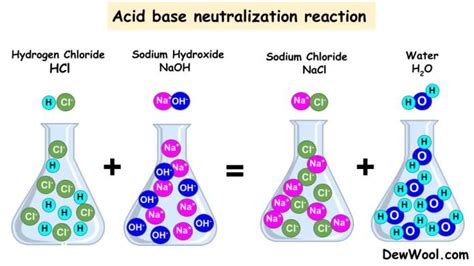
Table of Contents
Which Products Are the Result of a Neutralization Reaction?
Neutralization reactions are fundamental chemical processes with far-reaching implications in various industries and everyday life. Understanding these reactions and their resulting products is crucial for anyone interested in chemistry, materials science, or related fields. This comprehensive guide delves into the specifics of neutralization reactions, exploring the diverse products they yield and their applications.
Understanding Neutralization Reactions
A neutralization reaction, at its core, is a chemical reaction between an acid and a base. The reaction typically produces a salt and water. The defining characteristic is the combination of hydrogen ions (H⁺) from the acid and hydroxide ions (OH⁻) from the base to form water (H₂O). The remaining ions from the acid and base then combine to form a salt. The pH of the resulting solution is closer to 7 (neutral) than the original acidic or basic solution, hence the term "neutralization."
The General Equation:
Acid + Base → Salt + Water
This seemingly simple equation hides a vast array of possibilities, depending on the strength and type of acid and base involved. The nature of the salt produced significantly impacts the properties of the final product.
Strong Acid-Strong Base Neutralizations
Reactions between strong acids (like hydrochloric acid, HCl, or sulfuric acid, H₂SO₄) and strong bases (like sodium hydroxide, NaOH, or potassium hydroxide, KOH) are complete and readily proceed to completion. These reactions typically generate a neutral solution (pH 7) if stoichiometrically equivalent amounts of acid and base are used.
Example:
HCl (aq) + NaOH (aq) → NaCl (aq) + H₂O (l)
This reaction produces sodium chloride (table salt) and water. The resulting solution is neutral.
Weak Acid-Strong Base Neutralizations
When a weak acid (like acetic acid, CH₃COOH) reacts with a strong base, the resulting solution is slightly basic (pH > 7). This is because the conjugate base of the weak acid remains in solution and can react with water to produce hydroxide ions.
Example:
CH₃COOH (aq) + NaOH (aq) → CH₃COONa (aq) + H₂O (l)
This reaction produces sodium acetate and water. The sodium acetate, however, acts as a weak base, leading to a slightly alkaline solution.
Strong Acid-Weak Base Neutralizations
Conversely, a strong acid reacting with a weak base (like ammonia, NH₃) results in a slightly acidic solution (pH < 7). The conjugate acid of the weak base remains in solution and can react with water to produce hydronium ions.
Example:
HCl (aq) + NH₃ (aq) → NH₄Cl (aq)
This reaction produces ammonium chloride. The ammonium ion (NH₄⁺) acts as a weak acid, resulting in a slightly acidic solution.
Weak Acid-Weak Base Neutralizations
Reactions between weak acids and weak bases are more complex. The extent of the neutralization depends on the relative strengths of the acid and base. The resulting pH can vary significantly.
Diverse Products of Neutralization Reactions: A Detailed Look
The "salt" produced in a neutralization reaction is a broad category encompassing a wide range of chemical compounds. Their properties vary drastically depending on the acid and base involved. Let's explore some examples:
Salts: The Versatile Products
Salts are ionic compounds composed of a cation (positive ion) from the base and an anion (negative ion) from the acid. The properties of the salt, such as its solubility, melting point, and reactivity, depend on the specific ions involved.
1. Sodium Chloride (NaCl): As mentioned previously, the reaction between hydrochloric acid and sodium hydroxide produces common table salt. It's widely used in food preservation, seasoning, and various industrial processes.
2. Potassium Nitrate (KNO₃): Produced from the reaction of nitric acid and potassium hydroxide, potassium nitrate is a crucial component in fertilizers, providing nitrogen for plant growth. It also finds application in gunpowder and food preservation.
3. Calcium Carbonate (CaCO₃): While not directly a product of a simple acid-base neutralization, it's relevant to discuss its formation. Calcium carbonate, the primary component of limestone and marble, can be formed through precipitation reactions involving calcium ions and carbonate ions, which themselves can be generated through neutralization reactions involving calcium hydroxide and carbonic acid (a weak acid formed from carbon dioxide dissolving in water). This is crucial in understanding geological processes and the formation of various minerals.
4. Ammonium Sulfate ((NH₄)₂SO₄): This salt, produced from the reaction of sulfuric acid and ammonia, is a commonly used fertilizer providing both nitrogen and sulfur to plants.
5. Sodium Acetate (CH₃COONa): As discussed earlier, this salt is formed from the neutralization of acetic acid and sodium hydroxide. It's used as a buffer in chemical solutions, in photography, and as a food additive.
6. Copper Sulfate (CuSO₄): This salt, formed from the reaction of sulfuric acid and copper hydroxide, is bright blue in its hydrated form (CuSO₄·5H₂O) and has various applications, including fungicide, algaecide, and in electroplating.
Beyond Salts: Other Products
While salts and water are the primary products, other substances can form depending on the specific reactants and reaction conditions.
-
Gases: Certain neutralization reactions can produce gases, particularly if the acid or base is volatile or if a gas is involved in the reaction. For instance, the reaction of a carbonate with an acid produces carbon dioxide gas. CaCO₃(s) + 2HCl(aq) → CaCl₂(aq) + H₂O(l) + CO₂(g)
-
Precipitates: If the salt formed is insoluble in water, it precipitates out of the solution as a solid. This is important in various analytical techniques and industrial processes.
-
Complex Ions: In certain cases, complex ions can form, altering the properties of the resulting solution.
Applications of Neutralization Reactions and Their Products
The broad scope of neutralization reactions and their resulting products has led to their widespread use across various sectors:
-
Agriculture: Neutralization reactions are critical in soil management. The pH of soil needs to be within a certain range for optimal plant growth. Adding lime (calcium carbonate) to acidic soils neutralizes the acidity, creating a more favorable environment for plant roots. Fertilizers derived from neutralization reactions are indispensable for enhancing crop yields.
-
Medicine: Neutralization reactions play a critical role in treating acid indigestion. Antacids contain bases that neutralize excess stomach acid, providing relief from heartburn and indigestion.
-
Industry: Neutralization is crucial in various industrial processes. Wastewater treatment often involves neutralizing acidic or basic effluents before discharge into the environment. The production of numerous salts used in manufacturing, food processing, and other industries relies on neutralization reactions.
-
Environmental Science: Understanding neutralization reactions is essential for managing environmental pollution. Acid rain, resulting from the emission of acidic gases, can be neutralized using basic substances. Remediation of contaminated soil and water often involves neutralization strategies.
Conclusion: A Broad Chemical Landscape
Neutralization reactions represent a cornerstone of chemistry, underpinning a vast array of chemical processes and their applications. The products, primarily salts and water, exhibit remarkable diversity in properties and uses. From everyday applications like food seasoning and indigestion remedies to sophisticated industrial processes and environmental remediation strategies, neutralization reactions' influence is undeniable. The detailed understanding of these reactions and their resulting products is crucial for tackling various challenges in chemistry, materials science, and environmental management. Further research continues to uncover new applications and refine our understanding of these fundamental chemical processes.
Latest Posts
Latest Posts
-
What Is The Lcm Of 2 And 5
Apr 04, 2025
-
What Is 16 20 As A Percent
Apr 04, 2025
-
22 Is What Percent Of 40
Apr 04, 2025
-
Is Length An Intensive Or Extensive Property
Apr 04, 2025
-
14 Of 25 Is What Percent
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which Products Are The Result Of A Neutralization Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
