At What Temperature Does Water Evaporate
Kalali
Apr 05, 2025 · 6 min read
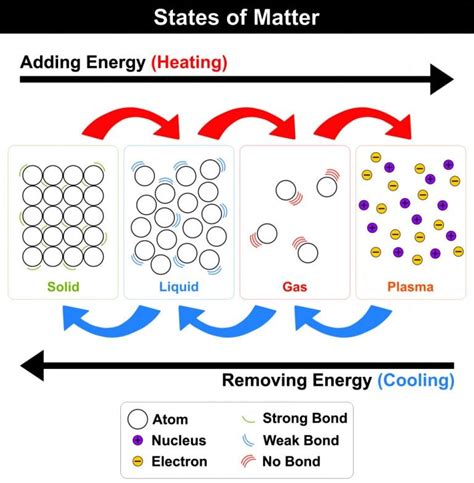
Table of Contents
At What Temperature Does Water Evaporate? Understanding the Science Behind Evaporation
Water, the elixir of life, is constantly in motion, undergoing a fascinating process called evaporation. This seemingly simple phenomenon is actually a complex interplay of temperature, pressure, and the water molecules themselves. Understanding at what temperature water evaporates requires delving into the world of thermodynamics and phase transitions. This comprehensive guide will explore the intricacies of water evaporation, dispelling common misconceptions and providing a thorough scientific explanation.
The Basics of Evaporation: More Than Just Boiling
Many mistakenly believe water only evaporates at its boiling point (100°C or 212°F at sea level). While boiling is a type of evaporation characterized by rapid vaporization throughout the liquid, evaporation itself is a much broader process. Evaporation is the transformation of water from its liquid state to its gaseous state (water vapor) at temperatures below its boiling point. This occurs continuously at any temperature above freezing (0°C or 32°F), albeit at varying rates.
The Role of Temperature
Temperature plays a crucial role in the rate of evaporation. Higher temperatures mean water molecules possess more kinetic energy. This increased energy allows them to overcome the intermolecular forces holding them together in the liquid phase, allowing them to escape into the atmosphere as water vapor. Conversely, lower temperatures result in slower evaporation as fewer molecules possess the energy to break free.
Beyond Temperature: Pressure and Humidity
While temperature is a primary driver, it's not the sole factor. Atmospheric pressure significantly impacts evaporation. Lower atmospheric pressure (like at higher altitudes) means less pressure pushing down on the water surface, making it easier for molecules to escape. This is why water boils at a lower temperature at higher altitudes.
Humidity, or the amount of water vapor already present in the air, also plays a critical role. Air can only hold a certain amount of water vapor at a given temperature; this is known as its saturation point. If the air is already saturated (high humidity), the rate of evaporation slows significantly, as there's less room for additional water vapor. Conversely, dry air (low humidity) allows for much faster evaporation.
The Molecular Dance: Understanding Water's Behavior
To fully grasp evaporation, we must examine the behavior of water molecules at a microscopic level. Water molecules are held together by relatively strong hydrogen bonds. These bonds create surface tension, influencing how easily molecules can escape the liquid's surface.
Kinetic Energy and Escape Velocity
At any temperature above freezing, water molecules are constantly moving and colliding. A small fraction of these molecules possess sufficient kinetic energy to overcome the hydrogen bonds and escape into the air. This energy is analogous to an "escape velocity" for the water molecules. The higher the temperature, the greater the proportion of molecules with this escape velocity.
Surface Area and Evaporation Rate
The surface area of the water body significantly impacts evaporation. A larger surface area exposes more water molecules to the atmosphere, leading to a higher evaporation rate. This is why a shallow dish of water evaporates faster than a deep container of the same volume. Think of it like this: more molecules have the opportunity to escape.
Factors Affecting Evaporation Rate: A Detailed Look
Numerous factors contribute to the rate of evaporation beyond temperature, pressure, and humidity. Understanding these nuances allows for a more precise prediction of evaporation rates in various scenarios.
Wind Speed
Wind plays a crucial role in evaporation. Moving air sweeps away water vapor molecules from the surface of the water, reducing the concentration of water vapor near the surface. This creates a concentration gradient, encouraging more molecules to escape the liquid phase to balance the gradient. High wind speeds result in faster evaporation.
Water Purity
The purity of water also affects its evaporation rate. Impurities such as dissolved salts can influence the intermolecular forces and slightly alter the boiling point. However, this effect is usually minor compared to the influence of temperature, pressure, and humidity.
Solar Radiation
Sunlight's energy directly increases the kinetic energy of water molecules, accelerating the evaporation process. Regions receiving high levels of solar radiation typically experience higher rates of evaporation.
Evaporation in Different Environments: Examples and Applications
Understanding the principles of evaporation is critical across various fields, from meteorology to engineering. Let's explore some practical examples.
Meteorology and Weather Patterns
Evaporation is a fundamental component of the water cycle, influencing weather patterns and climate. Evaporation from oceans, lakes, and rivers provides the atmospheric moisture that eventually forms clouds and precipitation. Accurate prediction of evaporation rates is vital for weather forecasting.
Agriculture and Irrigation
Understanding evaporation is crucial for effective irrigation management. Farmers need to account for evaporation losses when determining the amount of water needed for crops. Factors like temperature, humidity, and wind speed are carefully considered to optimize water usage and avoid water waste.
Industrial Processes
Many industrial processes utilize evaporation as a key step. For instance, desalination plants use evaporation to remove salt from seawater, providing a source of freshwater. Similarly, many food processing techniques rely on evaporation to concentrate solutions or dry products.
Human Body Temperature Regulation
Evaporation plays a critical role in regulating human body temperature. Sweating cools the body through evaporative heat loss. As sweat evaporates, it absorbs heat from the skin, lowering body temperature. This is particularly important during physical activity or in hot environments.
Misconceptions about Evaporation Debunked
Let's address some common misconceptions surrounding the evaporation process.
Myth 1: Evaporation only happens at 100°C (212°F). As discussed earlier, this is incorrect. Evaporation occurs at any temperature above freezing, albeit at varying rates. Boiling is a specific type of rapid evaporation that happens at the boiling point.
Myth 2: Evaporation is only affected by temperature. While temperature is a major factor, pressure, humidity, wind speed, surface area, and solar radiation all significantly impact the rate of evaporation.
Myth 3: All liquids evaporate at the same rate. The rate of evaporation is highly dependent on the specific liquid's properties, such as its boiling point, vapor pressure, and intermolecular forces.
Conclusion: A Deeper Understanding of Evaporation
Evaporation is a complex process driven by a multitude of interacting factors. While temperature is undeniably crucial, it's only one piece of the puzzle. Understanding the interplay of temperature, pressure, humidity, wind speed, surface area, and solar radiation allows for a more accurate prediction and control of evaporation rates across various applications. This comprehensive understanding is vital for progress in meteorology, agriculture, industry, and many other fields. By appreciating the intricate molecular dance underlying evaporation, we gain a deeper understanding of the world around us and the crucial role this process plays in shaping our environment.
Latest Posts
Latest Posts
-
8 Is What Percent Of 2
Apr 06, 2025
-
How Many Liters Are In 8 Gallons
Apr 06, 2025
-
What Is 32 Degrees Fahrenheit In Celsius
Apr 06, 2025
-
1 3 To The Power Of 3
Apr 06, 2025
-
What Is 30 Off Of 60
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about At What Temperature Does Water Evaporate . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
