Shapes Of Molecules Chart Chang Book
Kalali
Apr 04, 2025 · 6 min read
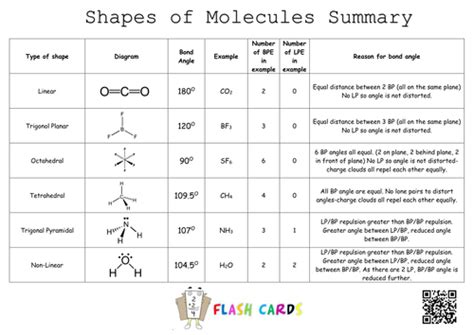
Table of Contents
Deciphering the Molecular World: A Deep Dive into Shapes of Molecules from Chang's Chemistry
Understanding the three-dimensional shapes of molecules is fundamental to comprehending their properties and reactivity. This is a concept thoroughly explored in numerous chemistry textbooks, notably in Kenneth L. Chang's widely used chemistry textbook. This article will delve deep into the subject of molecular shapes, drawing heavily on the principles presented in Chang's work, and providing a comprehensive guide to understanding this crucial aspect of chemistry. We'll explore various models for predicting molecular geometry, examine the influence of electron pairs (both bonding and non-bonding), and explore the consequences of different shapes on molecular polarity and properties.
The VSEPR Theory: A Cornerstone of Molecular Geometry Prediction
The Valence Shell Electron Pair Repulsion (VSEPR) theory forms the bedrock of our understanding of molecular shapes. Chang's textbook effectively introduces this theory, emphasizing the principle that electron pairs, both bonding and lone pairs, repel each other and arrange themselves to minimize this repulsion. This arrangement dictates the overall geometry of the molecule. The core concept is straightforward: electron pairs want to be as far apart as possible.
Predicting Molecular Shapes using VSEPR
To effectively utilize VSEPR, we follow a systematic approach:
-
Determine the Lewis Structure: The first step involves drawing the Lewis structure of the molecule. This gives us a clear picture of the bonding and non-bonding electrons.
-
Count the Electron Domains: An electron domain is defined as a region of high electron density. This includes both bonding pairs (single, double, or triple bonds are all counted as one domain) and lone pairs.
-
Determine the Electron Domain Geometry: The electron domain geometry describes the arrangement of electron domains around the central atom. Common geometries include linear (2 domains), trigonal planar (3 domains), tetrahedral (4 domains), trigonal bipyramidal (5 domains), and octahedral (6 domains).
-
Determine the Molecular Geometry: The molecular geometry considers only the positions of the atoms, ignoring the lone pairs. This is often different from the electron domain geometry, as lone pairs occupy more space than bonding pairs.
Examples of Molecular Shapes and their VSEPR Predictions
Let's illustrate the application of VSEPR with some examples frequently encountered in Chang's book and other introductory chemistry texts:
-
Linear: Molecules like BeCl₂ have two electron domains (two bonding pairs) and exhibit a linear geometry.
-
Bent (V-shaped): Water (H₂O) has four electron domains (two bonding pairs and two lone pairs). While the electron domain geometry is tetrahedral, the molecular geometry is bent due to the presence of lone pairs.
-
Trigonal Planar: Boron trifluoride (BF₃) displays a trigonal planar geometry with three electron domains (three bonding pairs) and no lone pairs.
-
Trigonal Pyramidal: Ammonia (NH₃) possesses four electron domains (three bonding pairs and one lone pair). The electron domain geometry is tetrahedral, but the molecular geometry is trigonal pyramidal.
-
Tetrahedral: Methane (CH₄) exemplifies a tetrahedral geometry with four electron domains (four bonding pairs) and no lone pairs.
-
Trigonal Bipyramidal & Octahedral: Molecules with five or six electron domains exhibit more complex geometries such as trigonal bipyramidal (e.g., PCl₅) and octahedral (e.g., SF₆). These geometries, often described in detail within Chang's comprehensive coverage, involve axial and equatorial positions for substituents, further influencing molecular properties.
Beyond VSEPR: Hybrid Orbitals and Molecular Orbital Theory
While VSEPR effectively predicts molecular shapes, a deeper understanding necessitates exploring hybrid orbital theory and molecular orbital theory. Chang's textbook likely introduces these concepts to provide a more complete picture of bonding and molecular structure.
Hybrid Orbital Theory
Hybrid orbital theory explains the observed geometries by combining atomic orbitals to form hybrid orbitals with different shapes and orientations. For example, the tetrahedral geometry of methane is explained by the hybridization of carbon's 2s and 2p orbitals to form four sp³ hybrid orbitals. Similarly, sp² hybridization explains the trigonal planar geometry, and sp hybridization accounts for linear geometries. This provides a more nuanced understanding of the electron distribution within the molecule.
Molecular Orbital Theory
Molecular orbital (MO) theory offers a more sophisticated approach. It describes bonding in terms of molecular orbitals formed by the combination of atomic orbitals. This theory accounts for phenomena like bond order and magnetic properties, which VSEPR cannot fully explain. Chang's treatment likely includes an overview of MO theory, perhaps focusing on simple diatomic molecules to illustrate the fundamental principles.
The Impact of Molecular Shape on Molecular Properties
The three-dimensional shape of a molecule profoundly influences its physical and chemical properties.
Polarity
The shape of a molecule dictates its polarity. Polar molecules have a dipole moment due to an uneven distribution of electron density. This arises from differences in electronegativity between atoms and the asymmetrical arrangement of these atoms. Symmetrical molecules like CO₂ are non-polar despite having polar bonds, while asymmetrical molecules like H₂O are polar even though the O-H bonds possess similar polarity.
Intermolecular Forces
Molecular shape significantly impacts intermolecular forces, which determine a substance's physical state (solid, liquid, or gas) and properties like boiling point and melting point. The strength of these forces (London dispersion forces, dipole-dipole interactions, hydrogen bonds) depends heavily on the molecule's shape and polarity. For instance, linear molecules can pack more efficiently, leading to stronger London dispersion forces compared to more branched structures.
Reactivity
Molecular shape also plays a vital role in chemical reactivity. The orientation of atoms in three-dimensional space determines how a molecule interacts with other molecules during chemical reactions. Specific functional groups, whose positions are determined by molecular geometry, influence the types of reactions a molecule undergoes. This aspect is crucial in organic chemistry and is often further expanded upon in more advanced texts building upon the fundamentals laid out by Chang.
Applications and Further Exploration
Understanding molecular shapes extends far beyond theoretical chemistry. It's crucial in various fields:
-
Drug Design: The shape of a drug molecule is critical for its interaction with receptor sites in the body. Drug designers meticulously manipulate molecular shapes to optimize efficacy and minimize side effects.
-
Materials Science: The properties of materials, whether polymers, ceramics, or metals, are strongly influenced by the shapes of their constituent molecules and how they interact.
-
Environmental Science: Understanding molecular shapes is essential for analyzing the behavior of pollutants and designing effective remediation strategies.
-
Biochemistry: Molecular shapes underpin the structure and function of biological macromolecules like proteins and DNA.
Chang's textbook likely provides a solid foundation for understanding these applications, offering examples and case studies that illustrate the practical relevance of molecular shapes.
Conclusion: Mastering Molecular Geometry with Chang
This article has provided a detailed exploration of molecular shapes, drawing inspiration from the principles laid out in Kenneth L. Chang's chemistry textbook. From the basic principles of VSEPR theory to the more advanced concepts of hybrid orbital and molecular orbital theories, understanding molecular geometry is paramount for comprehending the properties and reactivity of molecules. The influence of molecular shape on polarity, intermolecular forces, and reactivity underscores its importance across various scientific disciplines. By mastering this fundamental concept, you gain a powerful tool for interpreting and predicting the behavior of the molecular world. Further exploration of the topics discussed, along with the supplementary material provided within Chang's comprehensive text, will solidify your understanding and expand your capabilities in the field of chemistry. Remember, a thorough understanding of the principles presented here forms a vital stepping stone towards further exploration of more advanced chemical concepts.
Latest Posts
Latest Posts
-
What Is 102 Cm In Inches
Apr 05, 2025
-
What Is 58 Degrees Celsius In Fahrenheit
Apr 05, 2025
-
What Is 6 Percent Of 20
Apr 05, 2025
-
What Is 33 Inches In Feet
Apr 05, 2025
-
How To Know If An Quadratic Equation Is Linear
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Shapes Of Molecules Chart Chang Book . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
