What Is The Relationship Between Electron Affinity And Atomic Radius
Kalali
Apr 02, 2025 · 5 min read
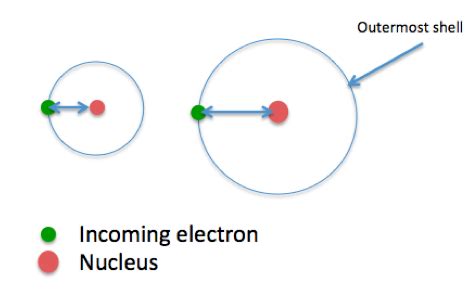
Table of Contents
The Intimate Dance of Electrons: Exploring the Relationship Between Electron Affinity and Atomic Radius
The world of atoms is a fascinating realm of intricate relationships, where seemingly simple properties intertwine in complex and predictable ways. Two such properties, electron affinity and atomic radius, are intrinsically linked, their dance revealing much about the fundamental behavior of matter. Understanding this relationship unlocks a deeper comprehension of chemical bonding, reactivity, and the periodic trends that govern the elements. This article delves into the intricacies of electron affinity and atomic radius, exploring their individual definitions, the nature of their relationship, and the exceptions that highlight the subtleties of atomic interactions.
Understanding Atomic Radius: The Size of an Atom
Before diving into the relationship, let's solidify our understanding of atomic radius. Atomic radius, simply put, is a measure of the size of an atom. However, defining this "size" isn't straightforward. Atoms don't have sharply defined boundaries like billiard balls. Instead, the electron cloud surrounding the nucleus gradually fades in density. Therefore, several definitions exist, each with its own context:
-
Covalent Radius: This is half the distance between the nuclei of two identical atoms bonded covalently. It's useful for comparing the sizes of atoms within molecules.
-
Metallic Radius: Half the distance between adjacent nuclei in a metallic crystal lattice. This definition applies specifically to metallic elements.
-
Van der Waals Radius: Half the distance between the nuclei of two identical, non-bonded atoms in close proximity. This is relevant when considering interactions between non-bonded atoms, such as in noble gases.
Regardless of the specific definition used, atomic radius generally increases down a group in the periodic table and decreases across a period (from left to right). The increase down a group is due to the addition of electron shells, pushing the outermost electrons further from the nucleus. The decrease across a period stems from the increasing nuclear charge pulling the electrons closer, despite the addition of electrons to the same shell.
Deciphering Electron Affinity: The Atom's Embrace of Electrons
Electron affinity (EA) is the energy change that occurs when an atom in the gaseous phase gains an electron. A positive electron affinity indicates that energy is released when an electron is added, making the process energetically favorable. Conversely, a negative electron affinity means that energy is absorbed, making the addition of an electron unfavorable. It's crucial to note that the sign convention can be confusing; a more negative value signifies a greater release of energy and thus a higher electron affinity.
The magnitude of electron affinity is influenced by several factors:
-
Nuclear Charge: A higher nuclear charge attracts electrons more strongly, leading to a more negative (higher) electron affinity.
-
Atomic Size: Smaller atoms have a stronger pull on incoming electrons, resulting in a more negative electron affinity. The added electron is closer to the nucleus, experiencing a stronger electrostatic attraction.
-
Electron Shielding: Inner electrons shield the outer electrons from the full nuclear charge. Increased shielding reduces the attraction for an added electron, leading to a less negative (lower) electron affinity.
-
Electron Configuration: Half-filled and completely filled subshells are relatively stable. Adding an electron to these configurations requires more energy, resulting in a less negative (lower) electron affinity, or even a positive value.
The Interplay: How Atomic Radius Impacts Electron Affinity
The relationship between atomic radius and electron affinity is largely inverse. As atomic radius increases, electron affinity generally decreases. This inverse relationship is a direct consequence of the electrostatic forces involved:
-
Distance and Attraction: A larger atomic radius means the added electron is further from the positively charged nucleus. This increased distance weakens the attractive force, making it less energetically favorable to add the electron, resulting in a less negative (or even positive) electron affinity.
-
Shielding Effect Enhancement: In larger atoms, the increased number of inner electrons enhances the shielding effect. This shielding reduces the net positive charge experienced by the incoming electron, further weakening the attraction and decreasing the electron affinity.
In essence, the smaller the atom, the greater its electron affinity (generally).
Exceptions and Nuances: The Complexity of Atomic Interactions
While the inverse relationship between atomic radius and electron affinity is generally true, exceptions exist. These exceptions highlight the intricate interplay of various factors influencing the electron affinity:
-
Electron-Electron Repulsion: Adding an electron to an atom already possessing a relatively large number of electrons can lead to increased electron-electron repulsion. This repulsion can outweigh the attractive force from the nucleus, potentially resulting in a less negative electron affinity than expected based solely on atomic radius.
-
Penetration Effects: The penetration of orbitals (s orbitals penetrate more than p orbitals) plays a role in determining the effective nuclear charge experienced by the added electron. This can introduce deviations from the simple inverse relationship.
-
Half-filled and Fully-filled Subshells: The exceptional stability of half-filled and completely filled subshells can significantly impact electron affinity. Adding an electron to a stable configuration requires more energy, leading to a less negative or even positive electron affinity. This is particularly evident in elements like nitrogen and oxygen, where the electron affinity trends deviate from the general pattern.
Practical Implications and Applications
Understanding the relationship between electron affinity and atomic radius has significant practical implications across several fields:
-
Predicting Chemical Reactivity: Elements with high electron affinities (small atomic radii) tend to be highly reactive, readily accepting electrons to form stable anions. This explains the high reactivity of halogens, for example.
-
Designing Materials: Knowledge of electron affinity is crucial in the design of new materials. It helps predict how atoms will interact, forming compounds with specific properties like conductivity or magnetism.
-
Understanding Biological Processes: Electron transfer reactions are fundamental to many biological processes. Understanding electron affinity helps decipher the mechanisms of these processes, such as respiration and photosynthesis.
Conclusion: A Deeper Look into Atomic Interactions
The relationship between electron affinity and atomic radius provides a valuable lens through which to examine the behavior of atoms. While a largely inverse relationship exists, exceptions highlight the complexity of atomic interactions. The interplay of nuclear charge, atomic size, shielding effects, and electron-electron repulsion paints a detailed picture of how atoms attract and interact with electrons, profoundly impacting their chemical properties and reactivity. A solid understanding of this relationship is essential for comprehending chemical bonding, materials science, and numerous biological phenomena. Further research continually refines our understanding of these atomic properties and their intricate dance, leading to advancements across various scientific and technological fields.
Latest Posts
Latest Posts
-
What Is 1 50 As A Percent
Apr 03, 2025
-
What Number Is 40 Percent Of 160
Apr 03, 2025
-
How Many Inches In 67 Cm
Apr 03, 2025
-
Which Are Two Ways A Population Can Decrease In Size
Apr 03, 2025
-
Which Products Are The Result Of A Neutralization Reaction
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Relationship Between Electron Affinity And Atomic Radius . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
