Classify Each Alcohol As A Primary Secondary Or Tertiary Alcohol
Kalali
Apr 05, 2025 · 5 min read
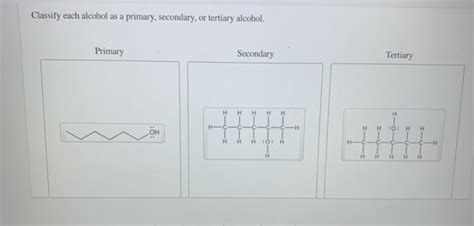
Table of Contents
Classifying Alcohols: Primary, Secondary, and Tertiary
Alcohols, a ubiquitous class of organic compounds, are characterized by the presence of a hydroxyl (-OH) group bonded to a carbon atom. Understanding the classification of alcohols—primary, secondary, or tertiary—is crucial for predicting their reactivity and understanding their properties. This comprehensive guide will delve into the intricacies of alcohol classification, exploring the structural basis, chemical reactions, and practical implications of each type.
Understanding the Fundamentals: The Carbon Skeleton
The key to classifying an alcohol lies in identifying the carbon atom to which the hydroxyl group is attached. This carbon atom's bonding environment—specifically, the number of other carbon atoms directly bonded to it—determines whether the alcohol is primary, secondary, or tertiary.
-
Primary Alcohols (1°): In a primary alcohol, the carbon atom bearing the hydroxyl group is bonded to only one other carbon atom. This means the carbon atom is either at the end of a carbon chain or part of a larger structure, but it only has one other carbon neighbor.
-
Secondary Alcohols (2°): A secondary alcohol features a hydroxyl group attached to a carbon atom bonded to two other carbon atoms. This carbon atom is usually found within a carbon chain.
-
Tertiary Alcohols (3°): In a tertiary alcohol, the hydroxyl-bearing carbon atom is connected to three other carbon atoms. This means it's typically found within a branched carbon chain or a more complex molecular structure.
Visualizing the Classification: Examples and Structures
Let's clarify the concepts with some examples, illustrating the structural differences between primary, secondary, and tertiary alcohols:
Primary Alcohol Examples:
-
Methanol (CH₃OH): The simplest alcohol, methanol, is a primary alcohol because the carbon atom attached to the hydroxyl group is bonded to only one other carbon atom (which is also bonded to three hydrogens).
-
Ethanol (CH₃CH₂OH): Ethanol, the alcohol in alcoholic beverages, is also a primary alcohol. The carbon bearing the -OH group is linked to only one other carbon atom.
-
1-Propanol (CH₃CH₂CH₂OH): 1-Propanol is another example; the hydroxyl group is attached to a terminal carbon, which only bonds to one other carbon.
Secondary Alcohol Examples:
-
2-Propanol (CH₃CH(OH)CH₃): Commonly known as isopropyl alcohol (rubbing alcohol), 2-propanol is a secondary alcohol. The carbon atom holding the -OH group is bonded to two other carbon atoms.
-
2-Butanol (CH₃CH₂CH(OH)CH₃): Similar to 2-propanol, 2-butanol has its hydroxyl group on a carbon atom bonded to two other carbons.
-
Cyclohexanol (C₆H₁₁OH): Cyclohexanol, an alcohol derived from cyclohexane, is secondary due to the hydroxyl group's attachment to a carbon atom bonded to two others within the ring structure.
Tertiary Alcohol Examples:
-
2-Methyl-2-propanol (tert-butyl alcohol): Often referred to as tert-butanol, this is a classic example of a tertiary alcohol. The carbon with the -OH is bonded to three other carbon atoms.
-
2-Methyl-2-butanol: This alcohol's hydroxyl group is on a carbon bonded to three others.
Chemical Reactivity: A Comparative Analysis
The classification of alcohols significantly influences their chemical reactivity. The different bonding environments around the hydroxyl-bearing carbon atom impact the stability of intermediates formed during reactions.
Oxidation Reactions: A Key Differentiator
Oxidation is a crucial reaction distinguishing primary, secondary, and tertiary alcohols. Using oxidizing agents like potassium dichromate (K₂Cr₂O₇) or potassium permanganate (KMnO₄), we observe distinct patterns:
-
Primary Alcohols: Primary alcohols undergo oxidation in two steps. The first step yields an aldehyde, and further oxidation converts the aldehyde to a carboxylic acid.
-
Secondary Alcohols: Secondary alcohols are oxidized to ketones. Ketones are less reactive than aldehydes and do not undergo further oxidation under typical conditions.
-
Tertiary Alcohols: Tertiary alcohols are resistant to oxidation under these conditions. The absence of a hydrogen atom on the hydroxyl-bearing carbon prevents the formation of the necessary intermediate for oxidation. Stronger oxidizing agents may cause cleavage of the C-C bonds, but this is a different type of reaction.
Other Reactions: Esterification and Dehydration
Besides oxidation, alcohols participate in other reactions where their classification influences the outcome:
-
Esterification: Alcohols react with carboxylic acids to form esters. While all three alcohol types can undergo esterification, the reaction rates and conditions might differ slightly.
-
Dehydration: The removal of a water molecule from an alcohol yields an alkene. The ease of dehydration and the resulting alkene products depend on the type of alcohol. Tertiary alcohols usually dehydrate more readily than secondary alcohols, which, in turn, dehydrate more readily than primary alcohols.
Practical Applications: Leveraging the Differences
The distinct properties and reactivities of primary, secondary, and tertiary alcohols lead to diverse applications in various fields:
-
Primary Alcohols: Methanol is used as a solvent and fuel. Ethanol finds applications as a solvent, fuel additive, and of course, in alcoholic beverages. Other primary alcohols are used as starting materials in the synthesis of various chemicals.
-
Secondary Alcohols: 2-Propanol (isopropyl alcohol) is a common disinfectant and solvent. Other secondary alcohols have applications in the pharmaceutical and chemical industries.
-
Tertiary Alcohols: Tertiary alcohols are often used as solvents or intermediates in organic synthesis. Their resistance to oxidation makes them useful in certain applications where stability is crucial.
Advanced Concepts and Considerations
While the basic classification of primary, secondary, and tertiary alcohols based on the carbon atom attached to the hydroxyl group is straightforward, some more nuanced aspects should be considered:
-
Polyols: Compounds with multiple hydroxyl groups (e.g., glycerol) fall outside the simple primary, secondary, tertiary categorization. Each hydroxyl group should be classified separately based on the carbon atom it's bonded to.
-
Cyclic Alcohols: Cyclic alcohols like cyclohexanol require careful consideration of the ring structure in determining the classification of the carbon atom bearing the hydroxyl group.
-
Chiral Alcohols: Some alcohols exhibit chirality, meaning they have non-superimposable mirror images. This chirality can impact their reactivity and physical properties.
Conclusion: Mastering Alcohol Classification
Understanding the classification of alcohols—primary, secondary, and tertiary—is fundamental to organic chemistry. The ability to classify alcohols accurately allows for predictions of their reactivity, which is critical in planning chemical syntheses and understanding their applications. This comprehensive guide provides a solid foundation for anyone studying or working with alcohols. Remember that the key lies in identifying the number of carbon atoms directly attached to the carbon atom bearing the hydroxyl (-OH) group. By mastering this simple rule, you can effectively navigate the complexities of alcohol chemistry and harness their diverse properties for various applications.
Latest Posts
Latest Posts
-
What Is Terminal Side Of The Angle
Apr 06, 2025
-
3 Kg Is Equal To How Many Grams
Apr 06, 2025
-
Cuanto Es Un Metro En Centimetros
Apr 06, 2025
-
25 Is What Percentage Of 75
Apr 06, 2025
-
Is Evaporation Exothermic Or Endothermic Process
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Classify Each Alcohol As A Primary Secondary Or Tertiary Alcohol . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
